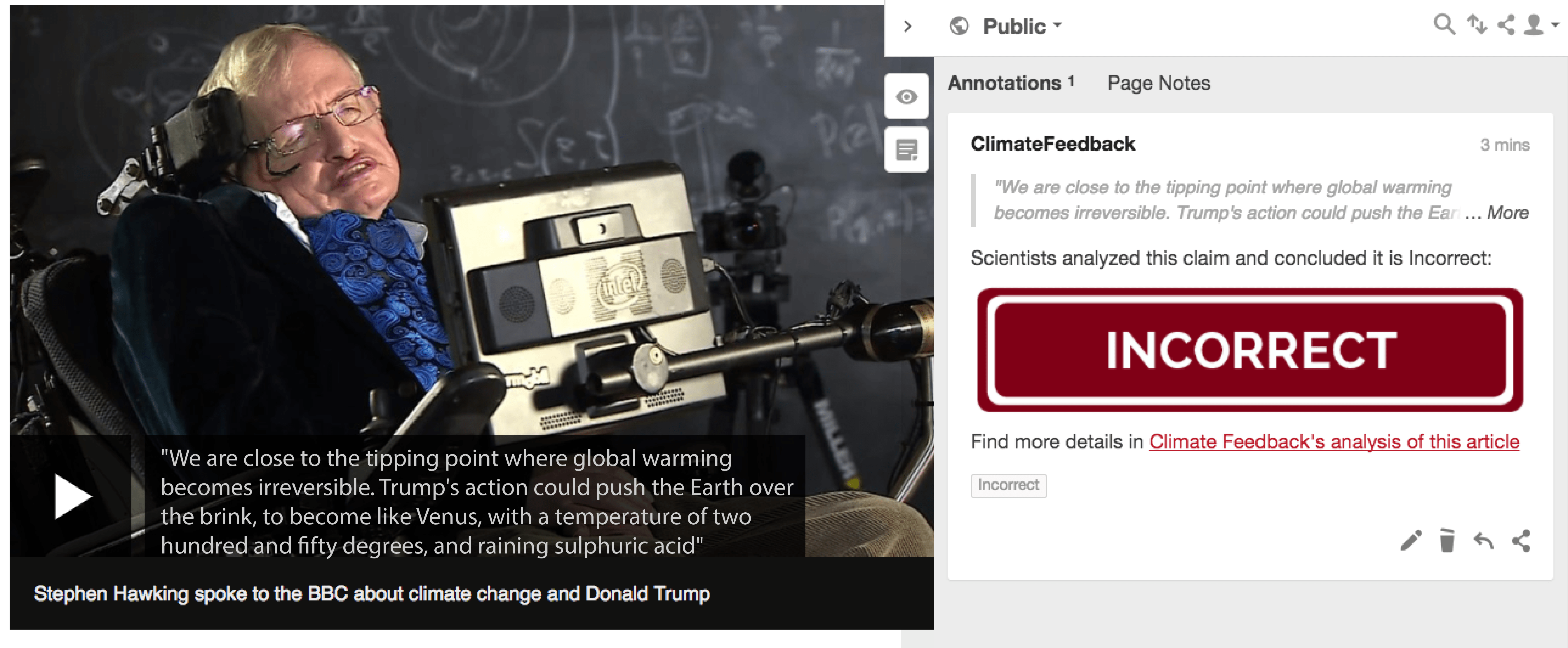
I was up to example #3 but cannot find the posts. I need help with that.
One of the examples involves the geologic carbon cycle including a Britannica article on the evolution of earth's atmosphere which quantified the amount of CO2 now trapped in earth's carbonates. Reposting the article link:
 www.britannica.com
www.britannica.com
The chart:
"Carbon in Earth's crust
form
total amount (Pg* C)
*One Pg (abbreviation for petagram) equals one quadrillion (1015) grams. Entries refer to amounts of carbon.
atmospheric CO (as of 1978)
696
oceanic carbon dioxide, bicarbonate ion, and carbonate ion
34,800
limestones, other carbonate sediments
64,800,000
carbonate in metamorphic rocks
2,640,000
total biomass
594
organic carbon in ocean water
996
organic carbon in soils
2,064
organic carbon in sedimentary rocks
12,000,000
organic carbon in metamorphic rocks
3,480,000
The table also emphasizes the dissolution of atmospheric gases by the ocean. The carbon dioxide in the atmosphere is in equilibrium with, and far less abundant than, the oceanic inventory of carbon dioxide, bicarbonate ions (HCO3−),"
Notice that most carbon in earth's crust is in the form of carbonates like limestone.
So - why didn't earth end up like Venus?
One of the examples involves the geologic carbon cycle including a Britannica article on the evolution of earth's atmosphere which quantified the amount of CO2 now trapped in earth's carbonates. Reposting the article link:

Evolution of the atmosphere - Biological Carbon Cycle
Evolution of the atmosphere - Biological Carbon Cycle: The biological processes of photosynthesis and respiration mediate the exchange of carbon between the atmosphere or hydrosphere and the biosphere, In these reactions, CH2O crudely represents organic material, the biomass of bacteria...
The chart:
"Carbon in Earth's crust
form
total amount (Pg* C)
*One Pg (abbreviation for petagram) equals one quadrillion (1015) grams. Entries refer to amounts of carbon.
atmospheric CO (as of 1978)
696
oceanic carbon dioxide, bicarbonate ion, and carbonate ion
34,800
limestones, other carbonate sediments
64,800,000
carbonate in metamorphic rocks
2,640,000
total biomass
594
organic carbon in ocean water
996
organic carbon in soils
2,064
organic carbon in sedimentary rocks
12,000,000
organic carbon in metamorphic rocks
3,480,000
The table also emphasizes the dissolution of atmospheric gases by the ocean. The carbon dioxide in the atmosphere is in equilibrium with, and far less abundant than, the oceanic inventory of carbon dioxide, bicarbonate ions (HCO3−),"
Notice that most carbon in earth's crust is in the form of carbonates like limestone.
So - why didn't earth end up like Venus?