Again, the link to the abstract (you need $40 to view the article):

 link.springer.com
link.springer.com
Excerpt:
""The first prebiotic electric discharge synthesis of amino acids showed that surprisingly high yields of amino acids were synthesized. Eleven amino acids were identified, four of which occur in proteins. Hydroxy acids, simple aliphatic acids and urea were also identified. These experiments have been repeated recently, and 33 amino acids were identified, ten of which occur in proteins, including all of the hydrophobic amino acids. ""
While this is relatively useless as I posted - one point is useful: the ratio of proteinous to non proteinous amino acids identified in the unidentified experiments. In the first case, 4 out of 11; in the second case 10 out of 33. This is consistent with the actual chemical reaction product proportions identified by Miller as per the chart (fig. 3-2) I am researching - I researched the top 10 in proportion, 3 of which are proteinous (found in proteins) - that is a ratio of 3 out of 10 - similar to the ratios in the above abstract quote.
Well here is where I left off:
Glycine - C₂H₅NO₂ - proportion: 440
Alanine - C3H7NO2 - proportion: 790
alpha-aminobutyric acid - C₄H₉NO₂ - proportion: 270
a(alpha)-Hydroxy-aminobutyric acid - C4H8O3 - proportion: 74
Norvaline - C5H11NO2 - proportion: 61
Sarcosine - C3H7NO2 - proportion: 55
Aspartic acid - C4H7NO4 - proportion: 34
2,4 [alpha/gamma]-diaminobutyric acid- C4H10N2O2 - proportion: 33
Note that only 3 of these (alanine, Glycine, aspartic acid) are among the 20 amino acids found in proteins - which are:
1. alanine - ala - A
2. arginine - arg - R
3. asparagine - asn - N
4. aspartic acid - asp - D
5. cysteine - cys - C
6. glutamine - gln - Q
7. glutamic acid - glu - E
8. glycine - gly - G
9. histidine - his - H
10. isoleucine - ile - I
11. leucine - leu - L
12. lysine - lys - K
13. methionine - met - M
14. phenylalanine - phe - F
15. proline - pro - P
16. serine - ser - S
17. threonine - thr - T
18. tryptophan - trp - W
19. tyrosine - tyr - Y
20. valine - val - V
So, now, back to my research:
The next 2 by proportion identified by Miller (proportion 30 for both) - from the chart here (p. 23, fig. 3-2) [see my previous posts on the first 8 by proportion]
a-Aminoisobutyric acid [a=alpha] - proportion 30 - from Bing search:
"2-Aminoisobutyric acid, or α-aminoisobutyric acid (AIB) or α-methylalanine or 2-methylalanine, is the non-proteinogenic amino acid with the structural formula H2N-C(CH3)2-COOH.....
chemical formula C₄H₉NO₂"
=============================
N-Ethylglycine
From:
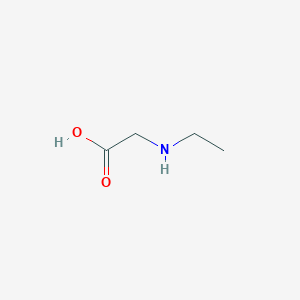
 pubchem.ncbi.nlm.nih.gov
pubchem.ncbi.nlm.nih.gov
= 2-(ethylamino)acetic acid
"N-ethylglycine is a N-alkylglycine. It is a tautomer of a N-ethylglycine zwitterion.....
N-ethylglycine belongs to the family of Alpha Amino Acids and Derivatives. These are amino acids in which the amino group is attached to the carbon atom immediately adjacent to the carboxylate group (alpha carbon)[1].....C4H9NO2"
A tautomer is an interchangeable isomer in equilibrium with at least one other isomer.
 en.wikipedia.org
en.wikipedia.org
"
Jump to search
A zwitterion (/ˈtsvɪtəˌraɪən/ TSVIT-ə-rye-ən; from German Zwitter [ˈtsvɪtɐ], meaning 'hermaphrodite') is a molecule that contains an equal number of positively-charged functional group(s) and negatively-charged functional group(s).[1] Zwitterions may also be called inner salts. With amino acids, for example, in solution a chemical equilibrium will be established between the "parent" molecule and the zwitterion.....
Amino acids[edit]

An amino acid contains both acidic (carboxylic acid fragment) and basic (amine fragment) centres. The isomer on the right is a zwitterion.
The zwitterion is formed in two stages. In stage 1, a proton is transferred from the carboxyl group to a water molecule.

In stage 2 a proton is transferred from the hydronium ion to the amine group

The reverse reactions occur in the reverse order. Overall, the reaction is an isomerization reaction
 "
"
I have already posted that numerous amino acids have numerous isomers (in this case tautomers).
N-Ethylglycine is one isomer/tautomer.
So N-Ethylglycine is not found in proteins (=non-proteinous/biologic) and was identified by Miller as relative proportion 30.

The atmosphere of the primitive Earth and the prebiotic synthesis of amino acids - Discover Life
The atmosphere of the Earth at the time of its formation is now generally believed to have been reducing, an idea proposed by Oparin and extensively discussed by Urey. This atmosphere would have contained CH4, N2 with traces of NH3, water and hydrogen. Only traces of NH3 would have been present...
Excerpt:
""The first prebiotic electric discharge synthesis of amino acids showed that surprisingly high yields of amino acids were synthesized. Eleven amino acids were identified, four of which occur in proteins. Hydroxy acids, simple aliphatic acids and urea were also identified. These experiments have been repeated recently, and 33 amino acids were identified, ten of which occur in proteins, including all of the hydrophobic amino acids. ""
While this is relatively useless as I posted - one point is useful: the ratio of proteinous to non proteinous amino acids identified in the unidentified experiments. In the first case, 4 out of 11; in the second case 10 out of 33. This is consistent with the actual chemical reaction product proportions identified by Miller as per the chart (fig. 3-2) I am researching - I researched the top 10 in proportion, 3 of which are proteinous (found in proteins) - that is a ratio of 3 out of 10 - similar to the ratios in the above abstract quote.
Well here is where I left off:
Glycine - C₂H₅NO₂ - proportion: 440
Alanine - C3H7NO2 - proportion: 790
alpha-aminobutyric acid - C₄H₉NO₂ - proportion: 270
a(alpha)-Hydroxy-aminobutyric acid - C4H8O3 - proportion: 74
Norvaline - C5H11NO2 - proportion: 61
Sarcosine - C3H7NO2 - proportion: 55
Aspartic acid - C4H7NO4 - proportion: 34
2,4 [alpha/gamma]-diaminobutyric acid- C4H10N2O2 - proportion: 33
Note that only 3 of these (alanine, Glycine, aspartic acid) are among the 20 amino acids found in proteins - which are:
1. alanine - ala - A
2. arginine - arg - R
3. asparagine - asn - N
4. aspartic acid - asp - D
5. cysteine - cys - C
6. glutamine - gln - Q
7. glutamic acid - glu - E
8. glycine - gly - G
9. histidine - his - H
10. isoleucine - ile - I
11. leucine - leu - L
12. lysine - lys - K
13. methionine - met - M
14. phenylalanine - phe - F
15. proline - pro - P
16. serine - ser - S
17. threonine - thr - T
18. tryptophan - trp - W
19. tyrosine - tyr - Y
20. valine - val - V
So, now, back to my research:
The next 2 by proportion identified by Miller (proportion 30 for both) - from the chart here (p. 23, fig. 3-2) [see my previous posts on the first 8 by proportion]
a-Aminoisobutyric acid [a=alpha] - proportion 30 - from Bing search:
"2-Aminoisobutyric acid, or α-aminoisobutyric acid (AIB) or α-methylalanine or 2-methylalanine, is the non-proteinogenic amino acid with the structural formula H2N-C(CH3)2-COOH.....
chemical formula C₄H₉NO₂"
=============================
N-Ethylglycine
From:

N-Ethylglycine
N-Ethylglycine | C4H9NO2 | CID 316542 - structure, chemical names, physical and chemical properties, classification, patents, literature, biological activities, safety/hazards/toxicity information, supplier lists, and more.
= 2-(ethylamino)acetic acid
"N-ethylglycine is a N-alkylglycine. It is a tautomer of a N-ethylglycine zwitterion.....
N-ethylglycine belongs to the family of Alpha Amino Acids and Derivatives. These are amino acids in which the amino group is attached to the carbon atom immediately adjacent to the carboxylate group (alpha carbon)[1].....C4H9NO2"
A tautomer is an interchangeable isomer in equilibrium with at least one other isomer.
Zwitterion - Wikipedia
"
Jump to search
A zwitterion (/ˈtsvɪtəˌraɪən/ TSVIT-ə-rye-ən; from German Zwitter [ˈtsvɪtɐ], meaning 'hermaphrodite') is a molecule that contains an equal number of positively-charged functional group(s) and negatively-charged functional group(s).[1] Zwitterions may also be called inner salts. With amino acids, for example, in solution a chemical equilibrium will be established between the "parent" molecule and the zwitterion.....
Amino acids[edit]

An amino acid contains both acidic (carboxylic acid fragment) and basic (amine fragment) centres. The isomer on the right is a zwitterion.
The zwitterion is formed in two stages. In stage 1, a proton is transferred from the carboxyl group to a water molecule.

In stage 2 a proton is transferred from the hydronium ion to the amine group

The reverse reactions occur in the reverse order. Overall, the reaction is an isomerization reaction

I have already posted that numerous amino acids have numerous isomers (in this case tautomers).
N-Ethylglycine is one isomer/tautomer.
So N-Ethylglycine is not found in proteins (=non-proteinous/biologic) and was identified by Miller as relative proportion 30.
 .
.