Amun Ra. Obviously."Before life began on Earth, the environment likely contained "As noted fatty acids were also produced in Miller's experiment (most sources ignore this). "
a carboxylic acid consisting of a hydrocarbon chain and a terminal carboxyl group ....
a fatty acid is a carboxylic acid with a long aliphatic chain, which is either saturated or unsaturated."
I posted above about carboxylic acids in general. The carboxyl group is a carbon atom double bonded to an Oxygen atom and single bonded to to a hydroxyl group (-OH). A hydrocarbon is a molecule with Hydrogen and Carbon (e.g. Methane = CH4). An aliphatic chain consists of hydrocarbons that form an open chain as in fatty acids and alkanes to be distinguished from hydrocarbon chains which form rings instead of open chains. Alkanes are saturated hydrocarbons like methane (CH4), ethane (C2H6) and propane (C3H8). Saturated hydrocarbons have single bonds between hydrogen atoms - the highest possible number of hydrogen atoms (e.g. methane/CH4) while unsaturated hydrocarbons have double or triple bonds between adjacent carbon atoms and thus can have hydrogen atoms added to make the compound saturated.
More from the Thaxton et al link concerning carboxylic acids from page 55:
"In a similar fashion, growing polypeptides would be terminated by reactions with amines, aldehydes, ketones, reducing sugars* or carboxylic acids. If by some remote chance a true protein did develop in the ocean, its viability would be predictably of short duration. For example, formaldehyde would readily react with free amino groups to form methylene cross-linkages between proteins.37 This would tie up certain reactive sites, and retard the reaction of protein with other chemical agents. To illustrate, "irreversible combination of formaldehyde with asparagine amide groups" would result in a compound which is "stable to dilute boiling phosphoric acid."38 This tying up process is the principle of the well-known tanning reaction, and is used similarly to retard cadaver decay. "In general, reaction with formaldehyde hardens proteins, decreases their water-sensitivity, and increases their resistance to the action of chemical reagents and enzymes."39 Survival of proteins in the soup would have been difficult indeed."
Footnote:
"It is interesting to note that in certain abnormal situations, such as diabetes, the carbonyl group of glucose will form chemical bonds with the amino group of cellular proteins, a process called glycosylation. (See A.L. Notkins, 1975. Sci. Amer. 241,62.)" [Not so good for life!]
Bottom line: carboxylic acids will react with peptides and stop further polymerization. Also the carbonyl group (CO double bonded) in sugars will react with the amino group (NH2) in peptides (and in amines in general). Both sugars (as in RNA) and amines (as in amino acids) are required for life and yet they interact and, in effect: destroy each other!
to be continued later. I'll give you all a chance to review the actual evidence in chemistry relevant to the origin of life and specifically to Miller's experiment.
Again, one needs an intelligent chemist (e.g. God) to create life - and human chemists are inferior creators and cannot create life. Some attribute the attributes of God to Chance! Not a chance!
Actually, we find that supernaturalism is not a requirement for life on the planet.
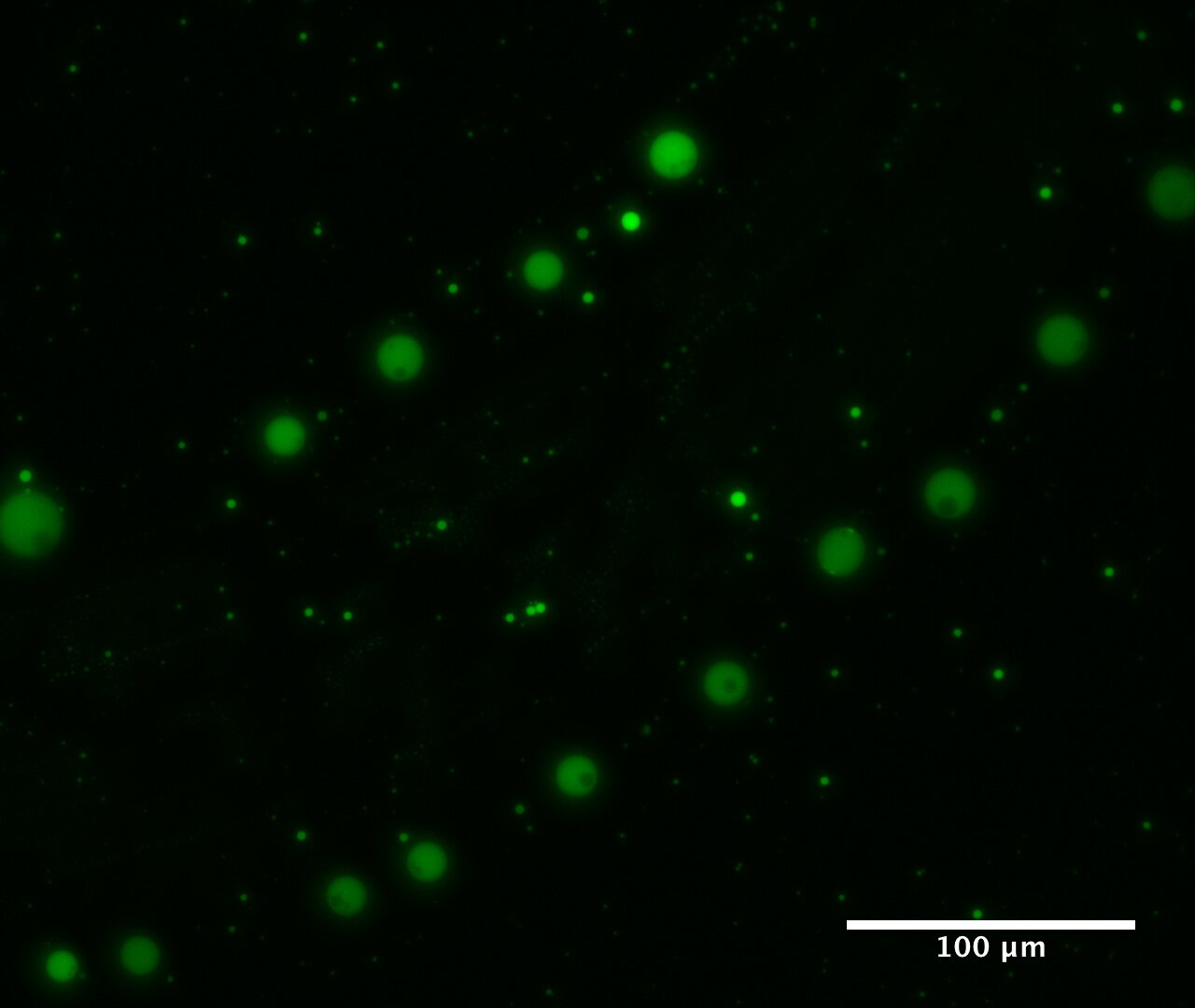
Before life began on Earth, the environment likely contained a massive number of chemicals that reacted with each other more or less randomly, and it is unclear how the complexity of cells could have emerged from such chemical chaos. Now, a team led by Tony Z. Jia at the Tokyo Institute of Technology and Kuhan Chandru of the National University of Malaysia has shown that simple α-hydroxy acids, like glycolic and lactic acid, spontaneously polymerize and self-assemble into polyester microdroplets when dried at moderate temperatures followed by rehydration. This could be what happened along primitive beaches and river banks, or in drying puddles. These form a new type of cell-like compartment that can trap and concentrate biomolecules like nucleic acids and proteins. These droplets, unlike most modern cells, are able to merge and reform easily, and thus could have hosted versatile early genetic and metabolic systems potentially critical for the origins of life.
All life on Earth is made up of cells. Cells are composed of lipids, proteins and nucleic acids, with the lipid forming the cell membrane, an enclosure that keeps the other components together and interfaces with the environment, exchanging food and waste. How molecular assemblages as complex as cells originally formed remains a mystery.
Most origins of life research focuses on how the molecules and structures of life were produced by the environment, and then assembled into structures that led to the first cells. However, there were likely many other types of molecules that formed alongside biomolecules on early Earth, and it is possible that life started using very simple chemistry unrelated to modern biomolecules, then evolved through increasingly complex stages to give rise to the structures found in modern cells.
Previous work conducted at ELSI showed that moderate temperature drying of the simple organic compounds known as alpha-hydroxy acids, which are found in meteorites and many simulations of prebiological chemistry, spontaneously polymerizes them into mixtures of long polyesters. Building on this work, Jia and colleagues took the next step and examined these reactions under the microscope, and found that these mixed polyester systems form a gel phase and spontaneously self-assemble when rewetted to form simple cell-like structures.
The most challenging aspect of this work was devising new methods to characterize the droplets' properties and functions, as no one had analyzed such systems before. Jia noted that the team was fortunate to have such a diversity of multidisciplinary expertise, including chemists, biochemists, materials scientists and geologists. After determining their composition and showing their propensity to self-assemble, the next question was whether these cell-like structures might be able to do something chemically useful. Modern cell membranes perform many crucial functions that help maintain the cell, for example, retaining macromolecules and metabolites in one place, as well as providing a constant internal environment, which can be very different from the one outside the cell. They first measured how stable these structures were and found they could persist for very long periods depending on the environmental conditions, but could also be made to merge and coalesce.
They then tested the ability of these structures to sequester molecules from the environment and found they accumulated large dye molecules to a remarkable degree. They then showed that these droplets could also host RNA and protein molecules and still permit them to be functionally catalytic. Further, the team showed that the droplets could assist in the formation of a lipid layer on their surface, suggesting they could have helped scaffold protocell formation.
Jia and colleagues are not certain these structures are the direct ancestors of cells, but they think it is possible such droplets could have enabled the assembly of protocells on Earth. The new compartmentalization system they have found is extremely simple, they note, and could form easily in primitive environments throughout the universe. Says Jia, "This allows us to imagine non-biological systems on early Earth that could still have had a hand in the origins of life. This suggests there may be many other non-biological systems that should be targets of future investigations of this type." He thinks the development of these or similar model systems could allow better study of the evolution of diverse chemical systems representative of the complex chemistries likely to be found on primitive planetary bodies.
"The early Earth was certainly a messy place chemically," Jia explains, "and often, most origins of life studies focus on modern biomolecules under relatively 'clean' conditions. Perhaps it is important to take these 'messy' mixtures and see if there are interesting functions or structures that can arise from them spontaneously." The authors now think that by systematically increasing the chemical complexity of such systems, they will be able to observe how they evolve over time and possibly discover divergent and emergent properties.
"We have this new experimental system we can now play with, so we can start to study phenomena like evolution and evolvability of these droplets. The possible combinations of structures or functions these droplets might have are almost endless. If the physical rules that govern the formation of droplets are fairly universal in nature, then we hope to study similar systems to discover whether they also can form microdroplets with novel properties," adds Jia.
Finally, while the team is presently focused on understanding the origins of life, they note this basic research could have applications in other areas, for example, drug delivery and personalized medicine. "This is just a wonderful example of the unexpected ways projects can develop when a team of diverse scientists from around the world come together to try and understand new and interesting phenomena," said team member Jim Cleaves, also of ELSI.
Scientists discover new chemistry that may help explain the origins of cellular life
Before life began on Earth, the environment likely contained a massive number of chemicals that reacted with each other more or less randomly, and it is unclear how the complexity of cells could have emerged from such chemical chaos. Now, a team led by Tony Z. Jia at the Tokyo Institute of...phys.org
Curious...Where did the environment come from?
Navigation
Install the app
How to install the app on iOS
Follow along with the video below to see how to install our site as a web app on your home screen.
Note: This feature may not be available in some browsers.
More options
You are using an out of date browser. It may not display this or other websites correctly.
You should upgrade or use an alternative browser.
You should upgrade or use an alternative browser.
Does God Exist?
- Thread starter Bruce Daniels
- Start date
Indeependent
Diamond Member
- Nov 19, 2013
- 73,633
- 28,506
- 2,250
Bowel cleasning?Amun Ra. Obviously."Before life began on Earth, the environment likely contained "As noted fatty acids were also produced in Miller's experiment (most sources ignore this). "
a carboxylic acid consisting of a hydrocarbon chain and a terminal carboxyl group ....
a fatty acid is a carboxylic acid with a long aliphatic chain, which is either saturated or unsaturated."
I posted above about carboxylic acids in general. The carboxyl group is a carbon atom double bonded to an Oxygen atom and single bonded to to a hydroxyl group (-OH). A hydrocarbon is a molecule with Hydrogen and Carbon (e.g. Methane = CH4). An aliphatic chain consists of hydrocarbons that form an open chain as in fatty acids and alkanes to be distinguished from hydrocarbon chains which form rings instead of open chains. Alkanes are saturated hydrocarbons like methane (CH4), ethane (C2H6) and propane (C3H8). Saturated hydrocarbons have single bonds between hydrogen atoms - the highest possible number of hydrogen atoms (e.g. methane/CH4) while unsaturated hydrocarbons have double or triple bonds between adjacent carbon atoms and thus can have hydrogen atoms added to make the compound saturated.
More from the Thaxton et al link concerning carboxylic acids from page 55:
"In a similar fashion, growing polypeptides would be terminated by reactions with amines, aldehydes, ketones, reducing sugars* or carboxylic acids. If by some remote chance a true protein did develop in the ocean, its viability would be predictably of short duration. For example, formaldehyde would readily react with free amino groups to form methylene cross-linkages between proteins.37 This would tie up certain reactive sites, and retard the reaction of protein with other chemical agents. To illustrate, "irreversible combination of formaldehyde with asparagine amide groups" would result in a compound which is "stable to dilute boiling phosphoric acid."38 This tying up process is the principle of the well-known tanning reaction, and is used similarly to retard cadaver decay. "In general, reaction with formaldehyde hardens proteins, decreases their water-sensitivity, and increases their resistance to the action of chemical reagents and enzymes."39 Survival of proteins in the soup would have been difficult indeed."
Footnote:
"It is interesting to note that in certain abnormal situations, such as diabetes, the carbonyl group of glucose will form chemical bonds with the amino group of cellular proteins, a process called glycosylation. (See A.L. Notkins, 1975. Sci. Amer. 241,62.)" [Not so good for life!]
Bottom line: carboxylic acids will react with peptides and stop further polymerization. Also the carbonyl group (CO double bonded) in sugars will react with the amino group (NH2) in peptides (and in amines in general). Both sugars (as in RNA) and amines (as in amino acids) are required for life and yet they interact and, in effect: destroy each other!
to be continued later. I'll give you all a chance to review the actual evidence in chemistry relevant to the origin of life and specifically to Miller's experiment.
Again, one needs an intelligent chemist (e.g. God) to create life - and human chemists are inferior creators and cannot create life. Some attribute the attributes of God to Chance! Not a chance!
Actually, we find that supernaturalism is not a requirement for life on the planet.
Before life began on Earth, the environment likely contained a massive number of chemicals that reacted with each other more or less randomly, and it is unclear how the complexity of cells could have emerged from such chemical chaos. Now, a team led by Tony Z. Jia at the Tokyo Institute of Technology and Kuhan Chandru of the National University of Malaysia has shown that simple α-hydroxy acids, like glycolic and lactic acid, spontaneously polymerize and self-assemble into polyester microdroplets when dried at moderate temperatures followed by rehydration. This could be what happened along primitive beaches and river banks, or in drying puddles. These form a new type of cell-like compartment that can trap and concentrate biomolecules like nucleic acids and proteins. These droplets, unlike most modern cells, are able to merge and reform easily, and thus could have hosted versatile early genetic and metabolic systems potentially critical for the origins of life.
All life on Earth is made up of cells. Cells are composed of lipids, proteins and nucleic acids, with the lipid forming the cell membrane, an enclosure that keeps the other components together and interfaces with the environment, exchanging food and waste. How molecular assemblages as complex as cells originally formed remains a mystery.
Most origins of life research focuses on how the molecules and structures of life were produced by the environment, and then assembled into structures that led to the first cells. However, there were likely many other types of molecules that formed alongside biomolecules on early Earth, and it is possible that life started using very simple chemistry unrelated to modern biomolecules, then evolved through increasingly complex stages to give rise to the structures found in modern cells.
Previous work conducted at ELSI showed that moderate temperature drying of the simple organic compounds known as alpha-hydroxy acids, which are found in meteorites and many simulations of prebiological chemistry, spontaneously polymerizes them into mixtures of long polyesters. Building on this work, Jia and colleagues took the next step and examined these reactions under the microscope, and found that these mixed polyester systems form a gel phase and spontaneously self-assemble when rewetted to form simple cell-like structures.
The most challenging aspect of this work was devising new methods to characterize the droplets' properties and functions, as no one had analyzed such systems before. Jia noted that the team was fortunate to have such a diversity of multidisciplinary expertise, including chemists, biochemists, materials scientists and geologists. After determining their composition and showing their propensity to self-assemble, the next question was whether these cell-like structures might be able to do something chemically useful. Modern cell membranes perform many crucial functions that help maintain the cell, for example, retaining macromolecules and metabolites in one place, as well as providing a constant internal environment, which can be very different from the one outside the cell. They first measured how stable these structures were and found they could persist for very long periods depending on the environmental conditions, but could also be made to merge and coalesce.
They then tested the ability of these structures to sequester molecules from the environment and found they accumulated large dye molecules to a remarkable degree. They then showed that these droplets could also host RNA and protein molecules and still permit them to be functionally catalytic. Further, the team showed that the droplets could assist in the formation of a lipid layer on their surface, suggesting they could have helped scaffold protocell formation.
Jia and colleagues are not certain these structures are the direct ancestors of cells, but they think it is possible such droplets could have enabled the assembly of protocells on Earth. The new compartmentalization system they have found is extremely simple, they note, and could form easily in primitive environments throughout the universe. Says Jia, "This allows us to imagine non-biological systems on early Earth that could still have had a hand in the origins of life. This suggests there may be many other non-biological systems that should be targets of future investigations of this type." He thinks the development of these or similar model systems could allow better study of the evolution of diverse chemical systems representative of the complex chemistries likely to be found on primitive planetary bodies.
"The early Earth was certainly a messy place chemically," Jia explains, "and often, most origins of life studies focus on modern biomolecules under relatively 'clean' conditions. Perhaps it is important to take these 'messy' mixtures and see if there are interesting functions or structures that can arise from them spontaneously." The authors now think that by systematically increasing the chemical complexity of such systems, they will be able to observe how they evolve over time and possibly discover divergent and emergent properties.
"We have this new experimental system we can now play with, so we can start to study phenomena like evolution and evolvability of these droplets. The possible combinations of structures or functions these droplets might have are almost endless. If the physical rules that govern the formation of droplets are fairly universal in nature, then we hope to study similar systems to discover whether they also can form microdroplets with novel properties," adds Jia.
Finally, while the team is presently focused on understanding the origins of life, they note this basic research could have applications in other areas, for example, drug delivery and personalized medicine. "This is just a wonderful example of the unexpected ways projects can develop when a team of diverse scientists from around the world come together to try and understand new and interesting phenomena," said team member Jim Cleaves, also of ELSI.

Scientists discover new chemistry that may help explain the origins of cellular life
Before life began on Earth, the environment likely contained a massive number of chemicals that reacted with each other more or less randomly, and it is unclear how the complexity of cells could have emerged from such chemical chaos. Now, a team led by Tony Z. Jia at the Tokyo Institute of...phys.org
Curious...Where did the environment come from?
Indeependent
Diamond Member
- Nov 19, 2013
- 73,633
- 28,506
- 2,250
Of course these studies omit how the atoms from which these atoms came about.Yet more evidence that creationists / supernaturalists are making appeals to gods who are not required for existence.

Spark of life: Metabolism appears in lab without cells
Complex processes that support all life have been serendipitously spotted with no cells needed, hinting that life could have started spontaneously in early oceanswww.newscientist.com
Metabolic processes that underpin life on Earth have arisen spontaneously outside of cells. The serendipitous finding that metabolism – the cascade of reactions in all cells that provides them with the raw materials they need to survive – can happen in such simple conditions provides fresh insights into how the first life formed. It also suggests that the complex processes needed for life may have surprisingly humble origins.
“People have said that these pathways look so complex they couldn’t form by environmental chemistry alone,” says Markus Ralser at the University of Cambridge who supervised the research.
But his findings suggest that many of these reactions could have occurred spontaneously in Earth’s early oceans, catalysed by metal ions rather than the enzymes that drive them in cells today.
The origin of metabolism is a major gap in our understanding of the emergence of life. “If you look at many different organisms from around the world, this network of reactions always looks very similar, suggesting that it must have come into place very early on in evolution, but no one knew precisely when or how,” says Ralser.
Happy accident
One theory is that RNA was the first building block of life because it helps to produce the enzymes that could catalyse complex sequences of reactions. Another possibility is that metabolism came first; perhaps even generating the molecules needed to make RNA, and that cells later incorporated these processes – but there was little evidence to support this.
“This is the first experiment showing that it is possible to create metabolic networks in the absence of RNA,” Ralser says.
Remarkably, the discovery was an accident, stumbled on during routine quality control testing of the medium used to culture cells at Ralser’s laboratory. As a shortcut, one of his students decided to run unused media through a mass spectrometer, which spotted a signal for pyruvate – an end product of a metabolic pathway called glycolysis.
To test whether the same processes could have helped spark life on Earth, they approached colleagues in the Earth sciences department who had been working on reconstructing the chemistry of the Archean Ocean, which covered the planet almost 4 billion years ago. This was an oxygen-free world, predating photosynthesis, when the waters were rich in iron, as well as other metals and phosphate. All these substances could potentially facilitate chemical reactions like the ones seen in modern cells.
Metabolic backbone
Ralser’s team took early ocean solutions and added substances known to be starting points for modern metabolic pathways, before heating the samples to between 50 ˚C and 70 ˚C – the sort of temperatures you might have found near a hydrothermal vent – for 5 hours. Ralser then analysed the solutions to see what molecules were present.
“In the beginning we had hoped to find one reaction or two maybe, but the results were amazing,” says Ralser. “We could reconstruct two metabolic pathways almost entirely.”
The pathways they detected were glycolysis and the pentose phosphate pathway, “reactions that form the core metabolic backbone of every living cell,” Ralser adds. Together these pathways produce some of the most important materials in modern cells, including ATP – the molecule cells use to drive their machinery, the sugars that form DNA and RNA, and the molecules needed to make fats and proteins.
If these metabolic pathways were occurring in the early oceans, then the first cells could have enveloped them as they developed membranes.
In all, 29 metabolism-like chemical reactions were spotted, seemingly catalysed by iron and other metals that would have been found in early ocean sediments. The metabolic pathways aren’t identical to modern ones; some of the chemicals made by intermediate steps weren’t detected. However, “if you compare them side by side it is the same structure and many of the same molecules are formed,” Ralser says. These pathways could have been refined and improved once enzymes evolved within cells.
Reversible reaction
Detecting the metabolite ribose 5-phosphate is particularly noteworthy, Ralser says. This is because it is a precursor to RNA, which encodes information, catalyses chemical reactions and most importantly of all, can replicate.
“I think this paper has really interesting connotations for the origins of life,” says Matthew Powner at University College London. It hints at how more complex enzymes could have evolved, he says, because substances that made these early processes more efficient would have been selected for.
There is one big problem, however. “For origins of life, it is important to understand where the source molecules come from,” Powner says. No one has yet shown that such substances could form spontaneously in the early oceans.
A related issue is that the reactions observed so far only go in one direction; from complex sugars to simpler molecules like pyruvate. “Given the data, one might well conclude that any organics in the ocean would have been totally degraded, rather than forming the basis of modern metabolism,” says Jack Szostak, who studies the origin of life at Harvard. “I would conclude that metabolism had to evolve, within cells, one reaction and one catalyst at a time.”
But Ralser disagrees. In his opinion, whether the reaction is catalysed by an enzyme or by a molecule in the Archean Ocean leads to the same result; “every chemical reaction is in principle reversible, whether an enzyme or a simple molecule is the catalyst,” he says.
Journal reference: Molecular Systems Biology, DOI: 10.1002/msb.20145228
Read more: Spark of life: Metabolism appears in lab without cells
How did the very first atom come about?
Science has a solution for your problem.Bowel cleasning?Amun Ra. Obviously."Before life began on Earth, the environment likely contained "As noted fatty acids were also produced in Miller's experiment (most sources ignore this). "
a carboxylic acid consisting of a hydrocarbon chain and a terminal carboxyl group ....
a fatty acid is a carboxylic acid with a long aliphatic chain, which is either saturated or unsaturated."
I posted above about carboxylic acids in general. The carboxyl group is a carbon atom double bonded to an Oxygen atom and single bonded to to a hydroxyl group (-OH). A hydrocarbon is a molecule with Hydrogen and Carbon (e.g. Methane = CH4). An aliphatic chain consists of hydrocarbons that form an open chain as in fatty acids and alkanes to be distinguished from hydrocarbon chains which form rings instead of open chains. Alkanes are saturated hydrocarbons like methane (CH4), ethane (C2H6) and propane (C3H8). Saturated hydrocarbons have single bonds between hydrogen atoms - the highest possible number of hydrogen atoms (e.g. methane/CH4) while unsaturated hydrocarbons have double or triple bonds between adjacent carbon atoms and thus can have hydrogen atoms added to make the compound saturated.
More from the Thaxton et al link concerning carboxylic acids from page 55:
"In a similar fashion, growing polypeptides would be terminated by reactions with amines, aldehydes, ketones, reducing sugars* or carboxylic acids. If by some remote chance a true protein did develop in the ocean, its viability would be predictably of short duration. For example, formaldehyde would readily react with free amino groups to form methylene cross-linkages between proteins.37 This would tie up certain reactive sites, and retard the reaction of protein with other chemical agents. To illustrate, "irreversible combination of formaldehyde with asparagine amide groups" would result in a compound which is "stable to dilute boiling phosphoric acid."38 This tying up process is the principle of the well-known tanning reaction, and is used similarly to retard cadaver decay. "In general, reaction with formaldehyde hardens proteins, decreases their water-sensitivity, and increases their resistance to the action of chemical reagents and enzymes."39 Survival of proteins in the soup would have been difficult indeed."
Footnote:
"It is interesting to note that in certain abnormal situations, such as diabetes, the carbonyl group of glucose will form chemical bonds with the amino group of cellular proteins, a process called glycosylation. (See A.L. Notkins, 1975. Sci. Amer. 241,62.)" [Not so good for life!]
Bottom line: carboxylic acids will react with peptides and stop further polymerization. Also the carbonyl group (CO double bonded) in sugars will react with the amino group (NH2) in peptides (and in amines in general). Both sugars (as in RNA) and amines (as in amino acids) are required for life and yet they interact and, in effect: destroy each other!
to be continued later. I'll give you all a chance to review the actual evidence in chemistry relevant to the origin of life and specifically to Miller's experiment.
Again, one needs an intelligent chemist (e.g. God) to create life - and human chemists are inferior creators and cannot create life. Some attribute the attributes of God to Chance! Not a chance!
Actually, we find that supernaturalism is not a requirement for life on the planet.
Before life began on Earth, the environment likely contained a massive number of chemicals that reacted with each other more or less randomly, and it is unclear how the complexity of cells could have emerged from such chemical chaos. Now, a team led by Tony Z. Jia at the Tokyo Institute of Technology and Kuhan Chandru of the National University of Malaysia has shown that simple α-hydroxy acids, like glycolic and lactic acid, spontaneously polymerize and self-assemble into polyester microdroplets when dried at moderate temperatures followed by rehydration. This could be what happened along primitive beaches and river banks, or in drying puddles. These form a new type of cell-like compartment that can trap and concentrate biomolecules like nucleic acids and proteins. These droplets, unlike most modern cells, are able to merge and reform easily, and thus could have hosted versatile early genetic and metabolic systems potentially critical for the origins of life.
All life on Earth is made up of cells. Cells are composed of lipids, proteins and nucleic acids, with the lipid forming the cell membrane, an enclosure that keeps the other components together and interfaces with the environment, exchanging food and waste. How molecular assemblages as complex as cells originally formed remains a mystery.
Most origins of life research focuses on how the molecules and structures of life were produced by the environment, and then assembled into structures that led to the first cells. However, there were likely many other types of molecules that formed alongside biomolecules on early Earth, and it is possible that life started using very simple chemistry unrelated to modern biomolecules, then evolved through increasingly complex stages to give rise to the structures found in modern cells.
Previous work conducted at ELSI showed that moderate temperature drying of the simple organic compounds known as alpha-hydroxy acids, which are found in meteorites and many simulations of prebiological chemistry, spontaneously polymerizes them into mixtures of long polyesters. Building on this work, Jia and colleagues took the next step and examined these reactions under the microscope, and found that these mixed polyester systems form a gel phase and spontaneously self-assemble when rewetted to form simple cell-like structures.
The most challenging aspect of this work was devising new methods to characterize the droplets' properties and functions, as no one had analyzed such systems before. Jia noted that the team was fortunate to have such a diversity of multidisciplinary expertise, including chemists, biochemists, materials scientists and geologists. After determining their composition and showing their propensity to self-assemble, the next question was whether these cell-like structures might be able to do something chemically useful. Modern cell membranes perform many crucial functions that help maintain the cell, for example, retaining macromolecules and metabolites in one place, as well as providing a constant internal environment, which can be very different from the one outside the cell. They first measured how stable these structures were and found they could persist for very long periods depending on the environmental conditions, but could also be made to merge and coalesce.
They then tested the ability of these structures to sequester molecules from the environment and found they accumulated large dye molecules to a remarkable degree. They then showed that these droplets could also host RNA and protein molecules and still permit them to be functionally catalytic. Further, the team showed that the droplets could assist in the formation of a lipid layer on their surface, suggesting they could have helped scaffold protocell formation.
Jia and colleagues are not certain these structures are the direct ancestors of cells, but they think it is possible such droplets could have enabled the assembly of protocells on Earth. The new compartmentalization system they have found is extremely simple, they note, and could form easily in primitive environments throughout the universe. Says Jia, "This allows us to imagine non-biological systems on early Earth that could still have had a hand in the origins of life. This suggests there may be many other non-biological systems that should be targets of future investigations of this type." He thinks the development of these or similar model systems could allow better study of the evolution of diverse chemical systems representative of the complex chemistries likely to be found on primitive planetary bodies.
"The early Earth was certainly a messy place chemically," Jia explains, "and often, most origins of life studies focus on modern biomolecules under relatively 'clean' conditions. Perhaps it is important to take these 'messy' mixtures and see if there are interesting functions or structures that can arise from them spontaneously." The authors now think that by systematically increasing the chemical complexity of such systems, they will be able to observe how they evolve over time and possibly discover divergent and emergent properties.
"We have this new experimental system we can now play with, so we can start to study phenomena like evolution and evolvability of these droplets. The possible combinations of structures or functions these droplets might have are almost endless. If the physical rules that govern the formation of droplets are fairly universal in nature, then we hope to study similar systems to discover whether they also can form microdroplets with novel properties," adds Jia.
Finally, while the team is presently focused on understanding the origins of life, they note this basic research could have applications in other areas, for example, drug delivery and personalized medicine. "This is just a wonderful example of the unexpected ways projects can develop when a team of diverse scientists from around the world come together to try and understand new and interesting phenomena," said team member Jim Cleaves, also of ELSI.

Scientists discover new chemistry that may help explain the origins of cellular life
Before life began on Earth, the environment likely contained a massive number of chemicals that reacted with each other more or less randomly, and it is unclear how the complexity of cells could have emerged from such chemical chaos. Now, a team led by Tony Z. Jia at the Tokyo Institute of...phys.org
Curious...Where did the environment come from?
The magic of the gods.Of course these studies omit how the atoms from which these atoms came about.Yet more evidence that creationists / supernaturalists are making appeals to gods who are not required for existence.

Spark of life: Metabolism appears in lab without cells
Complex processes that support all life have been serendipitously spotted with no cells needed, hinting that life could have started spontaneously in early oceanswww.newscientist.com
Metabolic processes that underpin life on Earth have arisen spontaneously outside of cells. The serendipitous finding that metabolism – the cascade of reactions in all cells that provides them with the raw materials they need to survive – can happen in such simple conditions provides fresh insights into how the first life formed. It also suggests that the complex processes needed for life may have surprisingly humble origins.
“People have said that these pathways look so complex they couldn’t form by environmental chemistry alone,” says Markus Ralser at the University of Cambridge who supervised the research.
But his findings suggest that many of these reactions could have occurred spontaneously in Earth’s early oceans, catalysed by metal ions rather than the enzymes that drive them in cells today.
The origin of metabolism is a major gap in our understanding of the emergence of life. “If you look at many different organisms from around the world, this network of reactions always looks very similar, suggesting that it must have come into place very early on in evolution, but no one knew precisely when or how,” says Ralser.
Happy accident
One theory is that RNA was the first building block of life because it helps to produce the enzymes that could catalyse complex sequences of reactions. Another possibility is that metabolism came first; perhaps even generating the molecules needed to make RNA, and that cells later incorporated these processes – but there was little evidence to support this.
“This is the first experiment showing that it is possible to create metabolic networks in the absence of RNA,” Ralser says.
Remarkably, the discovery was an accident, stumbled on during routine quality control testing of the medium used to culture cells at Ralser’s laboratory. As a shortcut, one of his students decided to run unused media through a mass spectrometer, which spotted a signal for pyruvate – an end product of a metabolic pathway called glycolysis.
To test whether the same processes could have helped spark life on Earth, they approached colleagues in the Earth sciences department who had been working on reconstructing the chemistry of the Archean Ocean, which covered the planet almost 4 billion years ago. This was an oxygen-free world, predating photosynthesis, when the waters were rich in iron, as well as other metals and phosphate. All these substances could potentially facilitate chemical reactions like the ones seen in modern cells.
Metabolic backbone
Ralser’s team took early ocean solutions and added substances known to be starting points for modern metabolic pathways, before heating the samples to between 50 ˚C and 70 ˚C – the sort of temperatures you might have found near a hydrothermal vent – for 5 hours. Ralser then analysed the solutions to see what molecules were present.
“In the beginning we had hoped to find one reaction or two maybe, but the results were amazing,” says Ralser. “We could reconstruct two metabolic pathways almost entirely.”
The pathways they detected were glycolysis and the pentose phosphate pathway, “reactions that form the core metabolic backbone of every living cell,” Ralser adds. Together these pathways produce some of the most important materials in modern cells, including ATP – the molecule cells use to drive their machinery, the sugars that form DNA and RNA, and the molecules needed to make fats and proteins.
If these metabolic pathways were occurring in the early oceans, then the first cells could have enveloped them as they developed membranes.
In all, 29 metabolism-like chemical reactions were spotted, seemingly catalysed by iron and other metals that would have been found in early ocean sediments. The metabolic pathways aren’t identical to modern ones; some of the chemicals made by intermediate steps weren’t detected. However, “if you compare them side by side it is the same structure and many of the same molecules are formed,” Ralser says. These pathways could have been refined and improved once enzymes evolved within cells.
Reversible reaction
Detecting the metabolite ribose 5-phosphate is particularly noteworthy, Ralser says. This is because it is a precursor to RNA, which encodes information, catalyses chemical reactions and most importantly of all, can replicate.
“I think this paper has really interesting connotations for the origins of life,” says Matthew Powner at University College London. It hints at how more complex enzymes could have evolved, he says, because substances that made these early processes more efficient would have been selected for.
There is one big problem, however. “For origins of life, it is important to understand where the source molecules come from,” Powner says. No one has yet shown that such substances could form spontaneously in the early oceans.
A related issue is that the reactions observed so far only go in one direction; from complex sugars to simpler molecules like pyruvate. “Given the data, one might well conclude that any organics in the ocean would have been totally degraded, rather than forming the basis of modern metabolism,” says Jack Szostak, who studies the origin of life at Harvard. “I would conclude that metabolism had to evolve, within cells, one reaction and one catalyst at a time.”
But Ralser disagrees. In his opinion, whether the reaction is catalysed by an enzyme or by a molecule in the Archean Ocean leads to the same result; “every chemical reaction is in principle reversible, whether an enzyme or a simple molecule is the catalyst,” he says.
Journal reference: Molecular Systems Biology, DOI: 10.1002/msb.20145228
Read more: Spark of life: Metabolism appears in lab without cells
How did the very first atom come about?
How did the very first gods come about?
As noted fatty acids were also produced in Miller's experiment (most sources ignore this). "
a carboxylic acid consisting of a hydrocarbon chain and a terminal carboxyl group ....
a fatty acid is a carboxylic acid with a long aliphatic chain, which is either saturated or unsaturated."
I posted above about carboxylic acids in general. The carboxyl group is a carbon atom double bonded to an Oxygen atom and single bonded to to a hydroxyl group (-OH). A hydrocarbon is a molecule with Hydrogen and Carbon (e.g. Methane = CH4). An aliphatic chain consists of hydrocarbons that form an open chain as in fatty acids and alkanes to be distinguished from hydrocarbon chains which form rings instead of open chains. Alkanes are saturated hydrocarbons like methane (CH4), ethane (C2H6) and propane (C3H8). Saturated hydrocarbons have single bonds between hydrogen atoms - the highest possible number of hydrogen atoms (e.g. methane/CH4) while unsaturated hydrocarbons have double or triple bonds between adjacent carbon atoms and thus can have hydrogen atoms added to make the compound saturated.
More from the Thaxton et al link concerning carboxylic acids from page 55:
"In a similar fashion, growing polypeptides would be terminated by reactions with amines, aldehydes, ketones, reducing sugars* or carboxylic acids. If by some remote chance a true protein did develop in the ocean, its viability would be predictably of short duration. For example, formaldehyde would readily react with free amino groups to form methylene cross-linkages between proteins.37 This would tie up certain reactive sites, and retard the reaction of protein with other chemical agents. To illustrate, "irreversible combination of formaldehyde with asparagine amide groups" would result in a compound which is "stable to dilute boiling phosphoric acid."38 This tying up process is the principle of the well-known tanning reaction, and is used similarly to retard cadaver decay. "In general, reaction with formaldehyde hardens proteins, decreases their water-sensitivity, and increases their resistance to the action of chemical reagents and enzymes."39 Survival of proteins in the soup would have been difficult indeed."
Footnote:
"It is interesting to note that in certain abnormal situations, such as diabetes, the carbonyl group of glucose will form chemical bonds with the amino group of cellular proteins, a process called glycosylation. (See A.L. Notkins, 1975. Sci. Amer. 241,62.)" [Not so good for life!]
Bottom line: carboxylic acids will react with peptides and stop further polymerization. Also the carbonyl group (CO double bonded) in sugars will react with the amino group (NH2) in peptides (and in amines in general). Both sugars (as in RNA) and amines (as in amino acids) are required for life and yet they interact and, in effect: destroy each other!
to be continued later. I'll give you all a chance to review the actual evidence in chemistry relevant to the origin of life and specifically to Miller's experiment.
Again, one needs an intelligent chemist (e.g. God) to create life - and human chemists are inferior creators and cannot create life. Some attribute the attributes of God to Chance! Not a chance!
Actually, we find that supernaturalism is not a requirement for life on the planet.
Before life began on Earth, the environment likely contained a massive number of chemicals that reacted with each other more or less randomly, and it is unclear how the complexity of cells could have emerged from such chemical chaos. Now, a team led by Tony Z. Jia at the Tokyo Institute of Technology and Kuhan Chandru of the National University of Malaysia has shown that simple α-hydroxy acids, like glycolic and lactic acid, spontaneously polymerize and self-assemble into polyester microdroplets when dried at moderate temperatures followed by rehydration. This could be what happened along primitive beaches and river banks, or in drying puddles. These form a new type of cell-like compartment that can trap and concentrate biomolecules like nucleic acids and proteins. These droplets, unlike most modern cells, are able to merge and reform easily, and thus could have hosted versatile early genetic and metabolic systems potentially critical for the origins of life.
All life on Earth is made up of cells. Cells are composed of lipids, proteins and nucleic acids, with the lipid forming the cell membrane, an enclosure that keeps the other components together and interfaces with the environment, exchanging food and waste. How molecular assemblages as complex as cells originally formed remains a mystery.
Most origins of life research focuses on how the molecules and structures of life were produced by the environment, and then assembled into structures that led to the first cells. However, there were likely many other types of molecules that formed alongside biomolecules on early Earth, and it is possible that life started using very simple chemistry unrelated to modern biomolecules, then evolved through increasingly complex stages to give rise to the structures found in modern cells.
Previous work conducted at ELSI showed that moderate temperature drying of the simple organic compounds known as alpha-hydroxy acids, which are found in meteorites and many simulations of prebiological chemistry, spontaneously polymerizes them into mixtures of long polyesters. Building on this work, Jia and colleagues took the next step and examined these reactions under the microscope, and found that these mixed polyester systems form a gel phase and spontaneously self-assemble when rewetted to form simple cell-like structures.
The most challenging aspect of this work was devising new methods to characterize the droplets' properties and functions, as no one had analyzed such systems before. Jia noted that the team was fortunate to have such a diversity of multidisciplinary expertise, including chemists, biochemists, materials scientists and geologists. After determining their composition and showing their propensity to self-assemble, the next question was whether these cell-like structures might be able to do something chemically useful. Modern cell membranes perform many crucial functions that help maintain the cell, for example, retaining macromolecules and metabolites in one place, as well as providing a constant internal environment, which can be very different from the one outside the cell. They first measured how stable these structures were and found they could persist for very long periods depending on the environmental conditions, but could also be made to merge and coalesce.
They then tested the ability of these structures to sequester molecules from the environment and found they accumulated large dye molecules to a remarkable degree. They then showed that these droplets could also host RNA and protein molecules and still permit them to be functionally catalytic. Further, the team showed that the droplets could assist in the formation of a lipid layer on their surface, suggesting they could have helped scaffold protocell formation.
Jia and colleagues are not certain these structures are the direct ancestors of cells, but they think it is possible such droplets could have enabled the assembly of protocells on Earth. The new compartmentalization system they have found is extremely simple, they note, and could form easily in primitive environments throughout the universe. Says Jia, "This allows us to imagine non-biological systems on early Earth that could still have had a hand in the origins of life. This suggests there may be many other non-biological systems that should be targets of future investigations of this type." He thinks the development of these or similar model systems could allow better study of the evolution of diverse chemical systems representative of the complex chemistries likely to be found on primitive planetary bodies.
"The early Earth was certainly a messy place chemically," Jia explains, "and often, most origins of life studies focus on modern biomolecules under relatively 'clean' conditions. Perhaps it is important to take these 'messy' mixtures and see if there are interesting functions or structures that can arise from them spontaneously." The authors now think that by systematically increasing the chemical complexity of such systems, they will be able to observe how they evolve over time and possibly discover divergent and emergent properties.
"We have this new experimental system we can now play with, so we can start to study phenomena like evolution and evolvability of these droplets. The possible combinations of structures or functions these droplets might have are almost endless. If the physical rules that govern the formation of droplets are fairly universal in nature, then we hope to study similar systems to discover whether they also can form microdroplets with novel properties," adds Jia.
Finally, while the team is presently focused on understanding the origins of life, they note this basic research could have applications in other areas, for example, drug delivery and personalized medicine. "This is just a wonderful example of the unexpected ways projects can develop when a team of diverse scientists from around the world come together to try and understand new and interesting phenomena," said team member Jim Cleaves, also of ELSI.

Scientists discover new chemistry that may help explain the origins of cellular life
Before life began on Earth, the environment likely contained a massive number of chemicals that reacted with each other more or less randomly, and it is unclear how the complexity of cells could have emerged from such chemical chaos. Now, a team led by Tony Z. Jia at the Tokyo Institute of...phys.org
Hollie - thank you for the link - it does not address the amino acids and proportions in Miller's experiment - in fact, it is research on how a cell membrane could have been synthesized, not how proteins could be created (human chemists LI cannot do this, btw).
Long cut and paste - did you understand any of it?
Your link notes:
"Cells are composed of lipids, proteins and nucleic acids, with the lipid forming the cell membrane, an enclosure that keeps the other components together and interfaces with the environment, exchanging food and waste. How molecular assemblages as complex as cells originally formed remains a mystery."
Note also that your link does not state details about the environment needed to form simple alpha-hydroxy acids nor the chemcal reactions required for their formation. And the membrane produced does not perform the complex functions actual living cells require to be living cells rather than dead cells.
Are you familiar with how complex the simplest living cell membranes are? Like how food and waste exchanging could be created in a cell membrane?
The skill and wisdom involved to create such a cell membrane requires a superior chemist. Not to mention forming the informational molecules necessary for life and getting them, and only them, inside this cell membrane enclosure!
I
I have no problem right now except that I haven't had breakfast yet.
What problem are you referring to?
Science has a solution for your problem.Bowel cleasning?Amun Ra. Obviously."Before life began on Earth, the environment likely contained "As noted fatty acids were also produced in Miller's experiment (most sources ignore this). "
a carboxylic acid consisting of a hydrocarbon chain and a terminal carboxyl group ....
a fatty acid is a carboxylic acid with a long aliphatic chain, which is either saturated or unsaturated."
I posted above about carboxylic acids in general. The carboxyl group is a carbon atom double bonded to an Oxygen atom and single bonded to to a hydroxyl group (-OH). A hydrocarbon is a molecule with Hydrogen and Carbon (e.g. Methane = CH4). An aliphatic chain consists of hydrocarbons that form an open chain as in fatty acids and alkanes to be distinguished from hydrocarbon chains which form rings instead of open chains. Alkanes are saturated hydrocarbons like methane (CH4), ethane (C2H6) and propane (C3H8). Saturated hydrocarbons have single bonds between hydrogen atoms - the highest possible number of hydrogen atoms (e.g. methane/CH4) while unsaturated hydrocarbons have double or triple bonds between adjacent carbon atoms and thus can have hydrogen atoms added to make the compound saturated.
More from the Thaxton et al link concerning carboxylic acids from page 55:
"In a similar fashion, growing polypeptides would be terminated by reactions with amines, aldehydes, ketones, reducing sugars* or carboxylic acids. If by some remote chance a true protein did develop in the ocean, its viability would be predictably of short duration. For example, formaldehyde would readily react with free amino groups to form methylene cross-linkages between proteins.37 This would tie up certain reactive sites, and retard the reaction of protein with other chemical agents. To illustrate, "irreversible combination of formaldehyde with asparagine amide groups" would result in a compound which is "stable to dilute boiling phosphoric acid."38 This tying up process is the principle of the well-known tanning reaction, and is used similarly to retard cadaver decay. "In general, reaction with formaldehyde hardens proteins, decreases their water-sensitivity, and increases their resistance to the action of chemical reagents and enzymes."39 Survival of proteins in the soup would have been difficult indeed."
Footnote:
"It is interesting to note that in certain abnormal situations, such as diabetes, the carbonyl group of glucose will form chemical bonds with the amino group of cellular proteins, a process called glycosylation. (See A.L. Notkins, 1975. Sci. Amer. 241,62.)" [Not so good for life!]
Bottom line: carboxylic acids will react with peptides and stop further polymerization. Also the carbonyl group (CO double bonded) in sugars will react with the amino group (NH2) in peptides (and in amines in general). Both sugars (as in RNA) and amines (as in amino acids) are required for life and yet they interact and, in effect: destroy each other!
to be continued later. I'll give you all a chance to review the actual evidence in chemistry relevant to the origin of life and specifically to Miller's experiment.
Again, one needs an intelligent chemist (e.g. God) to create life - and human chemists are inferior creators and cannot create life. Some attribute the attributes of God to Chance! Not a chance!
Actually, we find that supernaturalism is not a requirement for life on the planet.
Before life began on Earth, the environment likely contained a massive number of chemicals that reacted with each other more or less randomly, and it is unclear how the complexity of cells could have emerged from such chemical chaos. Now, a team led by Tony Z. Jia at the Tokyo Institute of Technology and Kuhan Chandru of the National University of Malaysia has shown that simple α-hydroxy acids, like glycolic and lactic acid, spontaneously polymerize and self-assemble into polyester microdroplets when dried at moderate temperatures followed by rehydration. This could be what happened along primitive beaches and river banks, or in drying puddles. These form a new type of cell-like compartment that can trap and concentrate biomolecules like nucleic acids and proteins. These droplets, unlike most modern cells, are able to merge and reform easily, and thus could have hosted versatile early genetic and metabolic systems potentially critical for the origins of life.
All life on Earth is made up of cells. Cells are composed of lipids, proteins and nucleic acids, with the lipid forming the cell membrane, an enclosure that keeps the other components together and interfaces with the environment, exchanging food and waste. How molecular assemblages as complex as cells originally formed remains a mystery.
Most origins of life research focuses on how the molecules and structures of life were produced by the environment, and then assembled into structures that led to the first cells. However, there were likely many other types of molecules that formed alongside biomolecules on early Earth, and it is possible that life started using very simple chemistry unrelated to modern biomolecules, then evolved through increasingly complex stages to give rise to the structures found in modern cells.
Previous work conducted at ELSI showed that moderate temperature drying of the simple organic compounds known as alpha-hydroxy acids, which are found in meteorites and many simulations of prebiological chemistry, spontaneously polymerizes them into mixtures of long polyesters. Building on this work, Jia and colleagues took the next step and examined these reactions under the microscope, and found that these mixed polyester systems form a gel phase and spontaneously self-assemble when rewetted to form simple cell-like structures.
The most challenging aspect of this work was devising new methods to characterize the droplets' properties and functions, as no one had analyzed such systems before. Jia noted that the team was fortunate to have such a diversity of multidisciplinary expertise, including chemists, biochemists, materials scientists and geologists. After determining their composition and showing their propensity to self-assemble, the next question was whether these cell-like structures might be able to do something chemically useful. Modern cell membranes perform many crucial functions that help maintain the cell, for example, retaining macromolecules and metabolites in one place, as well as providing a constant internal environment, which can be very different from the one outside the cell. They first measured how stable these structures were and found they could persist for very long periods depending on the environmental conditions, but could also be made to merge and coalesce.
They then tested the ability of these structures to sequester molecules from the environment and found they accumulated large dye molecules to a remarkable degree. They then showed that these droplets could also host RNA and protein molecules and still permit them to be functionally catalytic. Further, the team showed that the droplets could assist in the formation of a lipid layer on their surface, suggesting they could have helped scaffold protocell formation.
Jia and colleagues are not certain these structures are the direct ancestors of cells, but they think it is possible such droplets could have enabled the assembly of protocells on Earth. The new compartmentalization system they have found is extremely simple, they note, and could form easily in primitive environments throughout the universe. Says Jia, "This allows us to imagine non-biological systems on early Earth that could still have had a hand in the origins of life. This suggests there may be many other non-biological systems that should be targets of future investigations of this type." He thinks the development of these or similar model systems could allow better study of the evolution of diverse chemical systems representative of the complex chemistries likely to be found on primitive planetary bodies.
"The early Earth was certainly a messy place chemically," Jia explains, "and often, most origins of life studies focus on modern biomolecules under relatively 'clean' conditions. Perhaps it is important to take these 'messy' mixtures and see if there are interesting functions or structures that can arise from them spontaneously." The authors now think that by systematically increasing the chemical complexity of such systems, they will be able to observe how they evolve over time and possibly discover divergent and emergent properties.
"We have this new experimental system we can now play with, so we can start to study phenomena like evolution and evolvability of these droplets. The possible combinations of structures or functions these droplets might have are almost endless. If the physical rules that govern the formation of droplets are fairly universal in nature, then we hope to study similar systems to discover whether they also can form microdroplets with novel properties," adds Jia.
Finally, while the team is presently focused on understanding the origins of life, they note this basic research could have applications in other areas, for example, drug delivery and personalized medicine. "This is just a wonderful example of the unexpected ways projects can develop when a team of diverse scientists from around the world come together to try and understand new and interesting phenomena," said team member Jim Cleaves, also of ELSI.

Scientists discover new chemistry that may help explain the origins of cellular life
Before life began on Earth, the environment likely contained a massive number of chemicals that reacted with each other more or less randomly, and it is unclear how the complexity of cells could have emerged from such chemical chaos. Now, a team led by Tony Z. Jia at the Tokyo Institute of...phys.org
Curious...Where did the environment come from?
I have no problem right now except that I haven't had breakfast yet.
What problem are you referring to?
Indeependent
Diamond Member
- Nov 19, 2013
- 73,633
- 28,506
- 2,250
But you don't have any Links to these "solutions"?Science has a solution for your problem.Bowel cleasning?Amun Ra. Obviously."Before life began on Earth, the environment likely contained "As noted fatty acids were also produced in Miller's experiment (most sources ignore this). "
a carboxylic acid consisting of a hydrocarbon chain and a terminal carboxyl group ....
a fatty acid is a carboxylic acid with a long aliphatic chain, which is either saturated or unsaturated."
I posted above about carboxylic acids in general. The carboxyl group is a carbon atom double bonded to an Oxygen atom and single bonded to to a hydroxyl group (-OH). A hydrocarbon is a molecule with Hydrogen and Carbon (e.g. Methane = CH4). An aliphatic chain consists of hydrocarbons that form an open chain as in fatty acids and alkanes to be distinguished from hydrocarbon chains which form rings instead of open chains. Alkanes are saturated hydrocarbons like methane (CH4), ethane (C2H6) and propane (C3H8). Saturated hydrocarbons have single bonds between hydrogen atoms - the highest possible number of hydrogen atoms (e.g. methane/CH4) while unsaturated hydrocarbons have double or triple bonds between adjacent carbon atoms and thus can have hydrogen atoms added to make the compound saturated.
More from the Thaxton et al link concerning carboxylic acids from page 55:
"In a similar fashion, growing polypeptides would be terminated by reactions with amines, aldehydes, ketones, reducing sugars* or carboxylic acids. If by some remote chance a true protein did develop in the ocean, its viability would be predictably of short duration. For example, formaldehyde would readily react with free amino groups to form methylene cross-linkages between proteins.37 This would tie up certain reactive sites, and retard the reaction of protein with other chemical agents. To illustrate, "irreversible combination of formaldehyde with asparagine amide groups" would result in a compound which is "stable to dilute boiling phosphoric acid."38 This tying up process is the principle of the well-known tanning reaction, and is used similarly to retard cadaver decay. "In general, reaction with formaldehyde hardens proteins, decreases their water-sensitivity, and increases their resistance to the action of chemical reagents and enzymes."39 Survival of proteins in the soup would have been difficult indeed."
Footnote:
"It is interesting to note that in certain abnormal situations, such as diabetes, the carbonyl group of glucose will form chemical bonds with the amino group of cellular proteins, a process called glycosylation. (See A.L. Notkins, 1975. Sci. Amer. 241,62.)" [Not so good for life!]
Bottom line: carboxylic acids will react with peptides and stop further polymerization. Also the carbonyl group (CO double bonded) in sugars will react with the amino group (NH2) in peptides (and in amines in general). Both sugars (as in RNA) and amines (as in amino acids) are required for life and yet they interact and, in effect: destroy each other!
to be continued later. I'll give you all a chance to review the actual evidence in chemistry relevant to the origin of life and specifically to Miller's experiment.
Again, one needs an intelligent chemist (e.g. God) to create life - and human chemists are inferior creators and cannot create life. Some attribute the attributes of God to Chance! Not a chance!
Actually, we find that supernaturalism is not a requirement for life on the planet.
Before life began on Earth, the environment likely contained a massive number of chemicals that reacted with each other more or less randomly, and it is unclear how the complexity of cells could have emerged from such chemical chaos. Now, a team led by Tony Z. Jia at the Tokyo Institute of Technology and Kuhan Chandru of the National University of Malaysia has shown that simple α-hydroxy acids, like glycolic and lactic acid, spontaneously polymerize and self-assemble into polyester microdroplets when dried at moderate temperatures followed by rehydration. This could be what happened along primitive beaches and river banks, or in drying puddles. These form a new type of cell-like compartment that can trap and concentrate biomolecules like nucleic acids and proteins. These droplets, unlike most modern cells, are able to merge and reform easily, and thus could have hosted versatile early genetic and metabolic systems potentially critical for the origins of life.
All life on Earth is made up of cells. Cells are composed of lipids, proteins and nucleic acids, with the lipid forming the cell membrane, an enclosure that keeps the other components together and interfaces with the environment, exchanging food and waste. How molecular assemblages as complex as cells originally formed remains a mystery.
Most origins of life research focuses on how the molecules and structures of life were produced by the environment, and then assembled into structures that led to the first cells. However, there were likely many other types of molecules that formed alongside biomolecules on early Earth, and it is possible that life started using very simple chemistry unrelated to modern biomolecules, then evolved through increasingly complex stages to give rise to the structures found in modern cells.
Previous work conducted at ELSI showed that moderate temperature drying of the simple organic compounds known as alpha-hydroxy acids, which are found in meteorites and many simulations of prebiological chemistry, spontaneously polymerizes them into mixtures of long polyesters. Building on this work, Jia and colleagues took the next step and examined these reactions under the microscope, and found that these mixed polyester systems form a gel phase and spontaneously self-assemble when rewetted to form simple cell-like structures.
The most challenging aspect of this work was devising new methods to characterize the droplets' properties and functions, as no one had analyzed such systems before. Jia noted that the team was fortunate to have such a diversity of multidisciplinary expertise, including chemists, biochemists, materials scientists and geologists. After determining their composition and showing their propensity to self-assemble, the next question was whether these cell-like structures might be able to do something chemically useful. Modern cell membranes perform many crucial functions that help maintain the cell, for example, retaining macromolecules and metabolites in one place, as well as providing a constant internal environment, which can be very different from the one outside the cell. They first measured how stable these structures were and found they could persist for very long periods depending on the environmental conditions, but could also be made to merge and coalesce.
They then tested the ability of these structures to sequester molecules from the environment and found they accumulated large dye molecules to a remarkable degree. They then showed that these droplets could also host RNA and protein molecules and still permit them to be functionally catalytic. Further, the team showed that the droplets could assist in the formation of a lipid layer on their surface, suggesting they could have helped scaffold protocell formation.
Jia and colleagues are not certain these structures are the direct ancestors of cells, but they think it is possible such droplets could have enabled the assembly of protocells on Earth. The new compartmentalization system they have found is extremely simple, they note, and could form easily in primitive environments throughout the universe. Says Jia, "This allows us to imagine non-biological systems on early Earth that could still have had a hand in the origins of life. This suggests there may be many other non-biological systems that should be targets of future investigations of this type." He thinks the development of these or similar model systems could allow better study of the evolution of diverse chemical systems representative of the complex chemistries likely to be found on primitive planetary bodies.
"The early Earth was certainly a messy place chemically," Jia explains, "and often, most origins of life studies focus on modern biomolecules under relatively 'clean' conditions. Perhaps it is important to take these 'messy' mixtures and see if there are interesting functions or structures that can arise from them spontaneously." The authors now think that by systematically increasing the chemical complexity of such systems, they will be able to observe how they evolve over time and possibly discover divergent and emergent properties.
"We have this new experimental system we can now play with, so we can start to study phenomena like evolution and evolvability of these droplets. The possible combinations of structures or functions these droplets might have are almost endless. If the physical rules that govern the formation of droplets are fairly universal in nature, then we hope to study similar systems to discover whether they also can form microdroplets with novel properties," adds Jia.
Finally, while the team is presently focused on understanding the origins of life, they note this basic research could have applications in other areas, for example, drug delivery and personalized medicine. "This is just a wonderful example of the unexpected ways projects can develop when a team of diverse scientists from around the world come together to try and understand new and interesting phenomena," said team member Jim Cleaves, also of ELSI.

Scientists discover new chemistry that may help explain the origins of cellular life
Before life began on Earth, the environment likely contained a massive number of chemicals that reacted with each other more or less randomly, and it is unclear how the complexity of cells could have emerged from such chemical chaos. Now, a team led by Tony Z. Jia at the Tokyo Institute of...phys.org
Curious...Where did the environment come from?
Fact...Every scientist on YouTube, when asked where the first piece of matter came about, always answers, "It's almost as though a god created it".
By the way, the only time anyone in a scientists audience can even ask this blasphemous question is when an atheist scientist is debating an Orthodox Jewish scientist.
If this question comes up in from of any other audience the person is jeered, ridiculed and removed from the room.
Try asking a college professor and you'll get an F.
Indeependent
Diamond Member
- Nov 19, 2013
- 73,633
- 28,506
- 2,250
I don't believe in man made gods that need me to burn my child on a pyre.The magic of the gods.Of course these studies omit how the atoms from which these atoms came about.Yet more evidence that creationists / supernaturalists are making appeals to gods who are not required for existence.

Spark of life: Metabolism appears in lab without cells
Complex processes that support all life have been serendipitously spotted with no cells needed, hinting that life could have started spontaneously in early oceanswww.newscientist.com
Metabolic processes that underpin life on Earth have arisen spontaneously outside of cells. The serendipitous finding that metabolism – the cascade of reactions in all cells that provides them with the raw materials they need to survive – can happen in such simple conditions provides fresh insights into how the first life formed. It also suggests that the complex processes needed for life may have surprisingly humble origins.
“People have said that these pathways look so complex they couldn’t form by environmental chemistry alone,” says Markus Ralser at the University of Cambridge who supervised the research.
But his findings suggest that many of these reactions could have occurred spontaneously in Earth’s early oceans, catalysed by metal ions rather than the enzymes that drive them in cells today.
The origin of metabolism is a major gap in our understanding of the emergence of life. “If you look at many different organisms from around the world, this network of reactions always looks very similar, suggesting that it must have come into place very early on in evolution, but no one knew precisely when or how,” says Ralser.
Happy accident
One theory is that RNA was the first building block of life because it helps to produce the enzymes that could catalyse complex sequences of reactions. Another possibility is that metabolism came first; perhaps even generating the molecules needed to make RNA, and that cells later incorporated these processes – but there was little evidence to support this.
“This is the first experiment showing that it is possible to create metabolic networks in the absence of RNA,” Ralser says.
Remarkably, the discovery was an accident, stumbled on during routine quality control testing of the medium used to culture cells at Ralser’s laboratory. As a shortcut, one of his students decided to run unused media through a mass spectrometer, which spotted a signal for pyruvate – an end product of a metabolic pathway called glycolysis.
To test whether the same processes could have helped spark life on Earth, they approached colleagues in the Earth sciences department who had been working on reconstructing the chemistry of the Archean Ocean, which covered the planet almost 4 billion years ago. This was an oxygen-free world, predating photosynthesis, when the waters were rich in iron, as well as other metals and phosphate. All these substances could potentially facilitate chemical reactions like the ones seen in modern cells.
Metabolic backbone
Ralser’s team took early ocean solutions and added substances known to be starting points for modern metabolic pathways, before heating the samples to between 50 ˚C and 70 ˚C – the sort of temperatures you might have found near a hydrothermal vent – for 5 hours. Ralser then analysed the solutions to see what molecules were present.
“In the beginning we had hoped to find one reaction or two maybe, but the results were amazing,” says Ralser. “We could reconstruct two metabolic pathways almost entirely.”
The pathways they detected were glycolysis and the pentose phosphate pathway, “reactions that form the core metabolic backbone of every living cell,” Ralser adds. Together these pathways produce some of the most important materials in modern cells, including ATP – the molecule cells use to drive their machinery, the sugars that form DNA and RNA, and the molecules needed to make fats and proteins.
If these metabolic pathways were occurring in the early oceans, then the first cells could have enveloped them as they developed membranes.
In all, 29 metabolism-like chemical reactions were spotted, seemingly catalysed by iron and other metals that would have been found in early ocean sediments. The metabolic pathways aren’t identical to modern ones; some of the chemicals made by intermediate steps weren’t detected. However, “if you compare them side by side it is the same structure and many of the same molecules are formed,” Ralser says. These pathways could have been refined and improved once enzymes evolved within cells.
Reversible reaction
Detecting the metabolite ribose 5-phosphate is particularly noteworthy, Ralser says. This is because it is a precursor to RNA, which encodes information, catalyses chemical reactions and most importantly of all, can replicate.
“I think this paper has really interesting connotations for the origins of life,” says Matthew Powner at University College London. It hints at how more complex enzymes could have evolved, he says, because substances that made these early processes more efficient would have been selected for.
There is one big problem, however. “For origins of life, it is important to understand where the source molecules come from,” Powner says. No one has yet shown that such substances could form spontaneously in the early oceans.
A related issue is that the reactions observed so far only go in one direction; from complex sugars to simpler molecules like pyruvate. “Given the data, one might well conclude that any organics in the ocean would have been totally degraded, rather than forming the basis of modern metabolism,” says Jack Szostak, who studies the origin of life at Harvard. “I would conclude that metabolism had to evolve, within cells, one reaction and one catalyst at a time.”
But Ralser disagrees. In his opinion, whether the reaction is catalysed by an enzyme or by a molecule in the Archean Ocean leads to the same result; “every chemical reaction is in principle reversible, whether an enzyme or a simple molecule is the catalyst,” he says.
Journal reference: Molecular Systems Biology, DOI: 10.1002/msb.20145228
Read more: Spark of life: Metabolism appears in lab without cells
How did the very first atom come about?
How did the very first gods come about?
Nic
Nice bait and switch - you are an intelligent debater! So, do you believe in a first cause? Or an infinite number of causes and effects going back for infinite past time? I am posting about cause and effect required for the formation of proteins. Do you deny the scientific principle of cause and effect?
The magic of the gods.Of course these studies omit how the atoms from which these atoms came about.Yet more evidence that creationists / supernaturalists are making appeals to gods who are not required for existence.

Spark of life: Metabolism appears in lab without cells
Complex processes that support all life have been serendipitously spotted with no cells needed, hinting that life could have started spontaneously in early oceanswww.newscientist.com
Metabolic processes that underpin life on Earth have arisen spontaneously outside of cells. The serendipitous finding that metabolism – the cascade of reactions in all cells that provides them with the raw materials they need to survive – can happen in such simple conditions provides fresh insights into how the first life formed. It also suggests that the complex processes needed for life may have surprisingly humble origins.
“People have said that these pathways look so complex they couldn’t form by environmental chemistry alone,” says Markus Ralser at the University of Cambridge who supervised the research.
But his findings suggest that many of these reactions could have occurred spontaneously in Earth’s early oceans, catalysed by metal ions rather than the enzymes that drive them in cells today.
The origin of metabolism is a major gap in our understanding of the emergence of life. “If you look at many different organisms from around the world, this network of reactions always looks very similar, suggesting that it must have come into place very early on in evolution, but no one knew precisely when or how,” says Ralser.
Happy accident
One theory is that RNA was the first building block of life because it helps to produce the enzymes that could catalyse complex sequences of reactions. Another possibility is that metabolism came first; perhaps even generating the molecules needed to make RNA, and that cells later incorporated these processes – but there was little evidence to support this.
“This is the first experiment showing that it is possible to create metabolic networks in the absence of RNA,” Ralser says.
Remarkably, the discovery was an accident, stumbled on during routine quality control testing of the medium used to culture cells at Ralser’s laboratory. As a shortcut, one of his students decided to run unused media through a mass spectrometer, which spotted a signal for pyruvate – an end product of a metabolic pathway called glycolysis.
To test whether the same processes could have helped spark life on Earth, they approached colleagues in the Earth sciences department who had been working on reconstructing the chemistry of the Archean Ocean, which covered the planet almost 4 billion years ago. This was an oxygen-free world, predating photosynthesis, when the waters were rich in iron, as well as other metals and phosphate. All these substances could potentially facilitate chemical reactions like the ones seen in modern cells.
Metabolic backbone
Ralser’s team took early ocean solutions and added substances known to be starting points for modern metabolic pathways, before heating the samples to between 50 ˚C and 70 ˚C – the sort of temperatures you might have found near a hydrothermal vent – for 5 hours. Ralser then analysed the solutions to see what molecules were present.
“In the beginning we had hoped to find one reaction or two maybe, but the results were amazing,” says Ralser. “We could reconstruct two metabolic pathways almost entirely.”
The pathways they detected were glycolysis and the pentose phosphate pathway, “reactions that form the core metabolic backbone of every living cell,” Ralser adds. Together these pathways produce some of the most important materials in modern cells, including ATP – the molecule cells use to drive their machinery, the sugars that form DNA and RNA, and the molecules needed to make fats and proteins.
If these metabolic pathways were occurring in the early oceans, then the first cells could have enveloped them as they developed membranes.
In all, 29 metabolism-like chemical reactions were spotted, seemingly catalysed by iron and other metals that would have been found in early ocean sediments. The metabolic pathways aren’t identical to modern ones; some of the chemicals made by intermediate steps weren’t detected. However, “if you compare them side by side it is the same structure and many of the same molecules are formed,” Ralser says. These pathways could have been refined and improved once enzymes evolved within cells.
Reversible reaction
Detecting the metabolite ribose 5-phosphate is particularly noteworthy, Ralser says. This is because it is a precursor to RNA, which encodes information, catalyses chemical reactions and most importantly of all, can replicate.
“I think this paper has really interesting connotations for the origins of life,” says Matthew Powner at University College London. It hints at how more complex enzymes could have evolved, he says, because substances that made these early processes more efficient would have been selected for.
There is one big problem, however. “For origins of life, it is important to understand where the source molecules come from,” Powner says. No one has yet shown that such substances could form spontaneously in the early oceans.
A related issue is that the reactions observed so far only go in one direction; from complex sugars to simpler molecules like pyruvate. “Given the data, one might well conclude that any organics in the ocean would have been totally degraded, rather than forming the basis of modern metabolism,” says Jack Szostak, who studies the origin of life at Harvard. “I would conclude that metabolism had to evolve, within cells, one reaction and one catalyst at a time.”
But Ralser disagrees. In his opinion, whether the reaction is catalysed by an enzyme or by a molecule in the Archean Ocean leads to the same result; “every chemical reaction is in principle reversible, whether an enzyme or a simple molecule is the catalyst,” he says.
Journal reference: Molecular Systems Biology, DOI: 10.1002/msb.20145228
Read more: Spark of life: Metabolism appears in lab without cells
How did the very first atom come about?
How did the very first gods come about?
Nice bait and switch - you are an intelligent debater! So, do you believe in a first cause? Or an infinite number of causes and effects going back for infinite past time? I am posting about cause and effect required for the formation of proteins. Do you deny the scientific principle of cause and effect?
Me nei
Me neither. Good morning indeependent! Have you had your proteins yet?
I don't believe in man made gods that need me to burn my child on a pyre.The magic of the gods.Of course these studies omit how the atoms from which these atoms came about.Yet more evidence that creationists / supernaturalists are making appeals to gods who are not required for existence.

Spark of life: Metabolism appears in lab without cells
Complex processes that support all life have been serendipitously spotted with no cells needed, hinting that life could have started spontaneously in early oceanswww.newscientist.com
Metabolic processes that underpin life on Earth have arisen spontaneously outside of cells. The serendipitous finding that metabolism – the cascade of reactions in all cells that provides them with the raw materials they need to survive – can happen in such simple conditions provides fresh insights into how the first life formed. It also suggests that the complex processes needed for life may have surprisingly humble origins.
“People have said that these pathways look so complex they couldn’t form by environmental chemistry alone,” says Markus Ralser at the University of Cambridge who supervised the research.
But his findings suggest that many of these reactions could have occurred spontaneously in Earth’s early oceans, catalysed by metal ions rather than the enzymes that drive them in cells today.
The origin of metabolism is a major gap in our understanding of the emergence of life. “If you look at many different organisms from around the world, this network of reactions always looks very similar, suggesting that it must have come into place very early on in evolution, but no one knew precisely when or how,” says Ralser.
Happy accident
One theory is that RNA was the first building block of life because it helps to produce the enzymes that could catalyse complex sequences of reactions. Another possibility is that metabolism came first; perhaps even generating the molecules needed to make RNA, and that cells later incorporated these processes – but there was little evidence to support this.
“This is the first experiment showing that it is possible to create metabolic networks in the absence of RNA,” Ralser says.
Remarkably, the discovery was an accident, stumbled on during routine quality control testing of the medium used to culture cells at Ralser’s laboratory. As a shortcut, one of his students decided to run unused media through a mass spectrometer, which spotted a signal for pyruvate – an end product of a metabolic pathway called glycolysis.
To test whether the same processes could have helped spark life on Earth, they approached colleagues in the Earth sciences department who had been working on reconstructing the chemistry of the Archean Ocean, which covered the planet almost 4 billion years ago. This was an oxygen-free world, predating photosynthesis, when the waters were rich in iron, as well as other metals and phosphate. All these substances could potentially facilitate chemical reactions like the ones seen in modern cells.
Metabolic backbone
Ralser’s team took early ocean solutions and added substances known to be starting points for modern metabolic pathways, before heating the samples to between 50 ˚C and 70 ˚C – the sort of temperatures you might have found near a hydrothermal vent – for 5 hours. Ralser then analysed the solutions to see what molecules were present.
“In the beginning we had hoped to find one reaction or two maybe, but the results were amazing,” says Ralser. “We could reconstruct two metabolic pathways almost entirely.”
The pathways they detected were glycolysis and the pentose phosphate pathway, “reactions that form the core metabolic backbone of every living cell,” Ralser adds. Together these pathways produce some of the most important materials in modern cells, including ATP – the molecule cells use to drive their machinery, the sugars that form DNA and RNA, and the molecules needed to make fats and proteins.
If these metabolic pathways were occurring in the early oceans, then the first cells could have enveloped them as they developed membranes.
In all, 29 metabolism-like chemical reactions were spotted, seemingly catalysed by iron and other metals that would have been found in early ocean sediments. The metabolic pathways aren’t identical to modern ones; some of the chemicals made by intermediate steps weren’t detected. However, “if you compare them side by side it is the same structure and many of the same molecules are formed,” Ralser says. These pathways could have been refined and improved once enzymes evolved within cells.
Reversible reaction
Detecting the metabolite ribose 5-phosphate is particularly noteworthy, Ralser says. This is because it is a precursor to RNA, which encodes information, catalyses chemical reactions and most importantly of all, can replicate.
“I think this paper has really interesting connotations for the origins of life,” says Matthew Powner at University College London. It hints at how more complex enzymes could have evolved, he says, because substances that made these early processes more efficient would have been selected for.
There is one big problem, however. “For origins of life, it is important to understand where the source molecules come from,” Powner says. No one has yet shown that such substances could form spontaneously in the early oceans.
A related issue is that the reactions observed so far only go in one direction; from complex sugars to simpler molecules like pyruvate. “Given the data, one might well conclude that any organics in the ocean would have been totally degraded, rather than forming the basis of modern metabolism,” says Jack Szostak, who studies the origin of life at Harvard. “I would conclude that metabolism had to evolve, within cells, one reaction and one catalyst at a time.”
But Ralser disagrees. In his opinion, whether the reaction is catalysed by an enzyme or by a molecule in the Archean Ocean leads to the same result; “every chemical reaction is in principle reversible, whether an enzyme or a simple molecule is the catalyst,” he says.
Journal reference: Molecular Systems Biology, DOI: 10.1002/msb.20145228
Read more: Spark of life: Metabolism appears in lab without cells
How did the very first atom come about?
How did the very first gods come about?
Me neither. Good morning indeependent! Have you had your proteins yet?
Indeependent
Diamond Member
- Nov 19, 2013
- 73,633
- 28,506
- 2,250
My wife and I became vegetarians and we love our proteins!Me nei
I don't believe in man made gods that need me to burn my child on a pyre.The magic of the gods.Of course these studies omit how the atoms from which these atoms came about.Yet more evidence that creationists / supernaturalists are making appeals to gods who are not required for existence.

Spark of life: Metabolism appears in lab without cells
Complex processes that support all life have been serendipitously spotted with no cells needed, hinting that life could have started spontaneously in early oceanswww.newscientist.com
Metabolic processes that underpin life on Earth have arisen spontaneously outside of cells. The serendipitous finding that metabolism – the cascade of reactions in all cells that provides them with the raw materials they need to survive – can happen in such simple conditions provides fresh insights into how the first life formed. It also suggests that the complex processes needed for life may have surprisingly humble origins.
“People have said that these pathways look so complex they couldn’t form by environmental chemistry alone,” says Markus Ralser at the University of Cambridge who supervised the research.
But his findings suggest that many of these reactions could have occurred spontaneously in Earth’s early oceans, catalysed by metal ions rather than the enzymes that drive them in cells today.
The origin of metabolism is a major gap in our understanding of the emergence of life. “If you look at many different organisms from around the world, this network of reactions always looks very similar, suggesting that it must have come into place very early on in evolution, but no one knew precisely when or how,” says Ralser.
Happy accident
One theory is that RNA was the first building block of life because it helps to produce the enzymes that could catalyse complex sequences of reactions. Another possibility is that metabolism came first; perhaps even generating the molecules needed to make RNA, and that cells later incorporated these processes – but there was little evidence to support this.
“This is the first experiment showing that it is possible to create metabolic networks in the absence of RNA,” Ralser says.
Remarkably, the discovery was an accident, stumbled on during routine quality control testing of the medium used to culture cells at Ralser’s laboratory. As a shortcut, one of his students decided to run unused media through a mass spectrometer, which spotted a signal for pyruvate – an end product of a metabolic pathway called glycolysis.
To test whether the same processes could have helped spark life on Earth, they approached colleagues in the Earth sciences department who had been working on reconstructing the chemistry of the Archean Ocean, which covered the planet almost 4 billion years ago. This was an oxygen-free world, predating photosynthesis, when the waters were rich in iron, as well as other metals and phosphate. All these substances could potentially facilitate chemical reactions like the ones seen in modern cells.
Metabolic backbone
Ralser’s team took early ocean solutions and added substances known to be starting points for modern metabolic pathways, before heating the samples to between 50 ˚C and 70 ˚C – the sort of temperatures you might have found near a hydrothermal vent – for 5 hours. Ralser then analysed the solutions to see what molecules were present.
“In the beginning we had hoped to find one reaction or two maybe, but the results were amazing,” says Ralser. “We could reconstruct two metabolic pathways almost entirely.”
The pathways they detected were glycolysis and the pentose phosphate pathway, “reactions that form the core metabolic backbone of every living cell,” Ralser adds. Together these pathways produce some of the most important materials in modern cells, including ATP – the molecule cells use to drive their machinery, the sugars that form DNA and RNA, and the molecules needed to make fats and proteins.
If these metabolic pathways were occurring in the early oceans, then the first cells could have enveloped them as they developed membranes.
In all, 29 metabolism-like chemical reactions were spotted, seemingly catalysed by iron and other metals that would have been found in early ocean sediments. The metabolic pathways aren’t identical to modern ones; some of the chemicals made by intermediate steps weren’t detected. However, “if you compare them side by side it is the same structure and many of the same molecules are formed,” Ralser says. These pathways could have been refined and improved once enzymes evolved within cells.
Reversible reaction
Detecting the metabolite ribose 5-phosphate is particularly noteworthy, Ralser says. This is because it is a precursor to RNA, which encodes information, catalyses chemical reactions and most importantly of all, can replicate.
“I think this paper has really interesting connotations for the origins of life,” says Matthew Powner at University College London. It hints at how more complex enzymes could have evolved, he says, because substances that made these early processes more efficient would have been selected for.
There is one big problem, however. “For origins of life, it is important to understand where the source molecules come from,” Powner says. No one has yet shown that such substances could form spontaneously in the early oceans.
A related issue is that the reactions observed so far only go in one direction; from complex sugars to simpler molecules like pyruvate. “Given the data, one might well conclude that any organics in the ocean would have been totally degraded, rather than forming the basis of modern metabolism,” says Jack Szostak, who studies the origin of life at Harvard. “I would conclude that metabolism had to evolve, within cells, one reaction and one catalyst at a time.”
But Ralser disagrees. In his opinion, whether the reaction is catalysed by an enzyme or by a molecule in the Archean Ocean leads to the same result; “every chemical reaction is in principle reversible, whether an enzyme or a simple molecule is the catalyst,” he says.
Journal reference: Molecular Systems Biology, DOI: 10.1002/msb.20145228
Read more: Spark of life: Metabolism appears in lab without cells
How did the very first atom come about?
How did the very first gods come about?
Me neither. Good morning indeependent! Have you had your proteins yet?
From:

 en.wikipedia.org
en.wikipedia.org
" Urey and Miller designed to simulate the ocean-atmospheric condition of the primitive Earth by using a continuous run of steam into a mixture of methane (CH4), ammonia (NH3), and Oxygen (O2). The gaseous mixture was then exposed to electrical discharge, which induced chemical reaction. After a week of reaction, Miller detected the formation of amino acids, such as glycine, α- and β-alanine, using paper chromatography. He also detected aspartic acid and gamma-amino butyric acid, but was not confident because of the weak spots."
A glaring error is saying Oxygen was used by Miller in his experiment - that should read Hydrogen. Oxygen would have destroyed any chemical reaction leading to HCN and all the other steps required to produce a polypeptide.
The rest is fairly accurate although limited and biased. For example, it is true that both alpha and beta Alanine were synthesized but the fact that only alpha alanine is used in proteins is not stated. It is also not noted that gamma-amino butyric acid is not in proteins.
I have already posted the chemical reaction product proportions reported by Miller - I will repost that in my next post. Not sure why wikipedia reports on gamma-aminobutyric acid since it is only in proportion 2.4 compared with alpha gamma Diaminobutyric acid with a proportion of 33 and alpha hydroxy gamma butyric acid with a proportion of 74 (both in the top 10 amino acids produced by Miller in order of proportion).

Stanley Miller - Wikipedia
 en.wikipedia.org
en.wikipedia.org
" Urey and Miller designed to simulate the ocean-atmospheric condition of the primitive Earth by using a continuous run of steam into a mixture of methane (CH4), ammonia (NH3), and Oxygen (O2). The gaseous mixture was then exposed to electrical discharge, which induced chemical reaction. After a week of reaction, Miller detected the formation of amino acids, such as glycine, α- and β-alanine, using paper chromatography. He also detected aspartic acid and gamma-amino butyric acid, but was not confident because of the weak spots."
A glaring error is saying Oxygen was used by Miller in his experiment - that should read Hydrogen. Oxygen would have destroyed any chemical reaction leading to HCN and all the other steps required to produce a polypeptide.
The rest is fairly accurate although limited and biased. For example, it is true that both alpha and beta Alanine were synthesized but the fact that only alpha alanine is used in proteins is not stated. It is also not noted that gamma-amino butyric acid is not in proteins.
I have already posted the chemical reaction product proportions reported by Miller - I will repost that in my next post. Not sure why wikipedia reports on gamma-aminobutyric acid since it is only in proportion 2.4 compared with alpha gamma Diaminobutyric acid with a proportion of 33 and alpha hydroxy gamma butyric acid with a proportion of 74 (both in the top 10 amino acids produced by Miller in order of proportion).
As in rice and beans? So much better than the cyanide (HCN) that Miller produced.My wife and I became vegetarians and we love our proteins!Me nei
I don't believe in man made gods that need me to burn my child on a pyre.The magic of the gods.Of course these studies omit how the atoms from which these atoms came about.Yet more evidence that creationists / supernaturalists are making appeals to gods who are not required for existence.

Spark of life: Metabolism appears in lab without cells
Complex processes that support all life have been serendipitously spotted with no cells needed, hinting that life could have started spontaneously in early oceanswww.newscientist.com
Metabolic processes that underpin life on Earth have arisen spontaneously outside of cells. The serendipitous finding that metabolism – the cascade of reactions in all cells that provides them with the raw materials they need to survive – can happen in such simple conditions provides fresh insights into how the first life formed. It also suggests that the complex processes needed for life may have surprisingly humble origins.
“People have said that these pathways look so complex they couldn’t form by environmental chemistry alone,” says Markus Ralser at the University of Cambridge who supervised the research.
But his findings suggest that many of these reactions could have occurred spontaneously in Earth’s early oceans, catalysed by metal ions rather than the enzymes that drive them in cells today.
The origin of metabolism is a major gap in our understanding of the emergence of life. “If you look at many different organisms from around the world, this network of reactions always looks very similar, suggesting that it must have come into place very early on in evolution, but no one knew precisely when or how,” says Ralser.
Happy accident
One theory is that RNA was the first building block of life because it helps to produce the enzymes that could catalyse complex sequences of reactions. Another possibility is that metabolism came first; perhaps even generating the molecules needed to make RNA, and that cells later incorporated these processes – but there was little evidence to support this.
“This is the first experiment showing that it is possible to create metabolic networks in the absence of RNA,” Ralser says.
Remarkably, the discovery was an accident, stumbled on during routine quality control testing of the medium used to culture cells at Ralser’s laboratory. As a shortcut, one of his students decided to run unused media through a mass spectrometer, which spotted a signal for pyruvate – an end product of a metabolic pathway called glycolysis.
To test whether the same processes could have helped spark life on Earth, they approached colleagues in the Earth sciences department who had been working on reconstructing the chemistry of the Archean Ocean, which covered the planet almost 4 billion years ago. This was an oxygen-free world, predating photosynthesis, when the waters were rich in iron, as well as other metals and phosphate. All these substances could potentially facilitate chemical reactions like the ones seen in modern cells.
Metabolic backbone
Ralser’s team took early ocean solutions and added substances known to be starting points for modern metabolic pathways, before heating the samples to between 50 ˚C and 70 ˚C – the sort of temperatures you might have found near a hydrothermal vent – for 5 hours. Ralser then analysed the solutions to see what molecules were present.
“In the beginning we had hoped to find one reaction or two maybe, but the results were amazing,” says Ralser. “We could reconstruct two metabolic pathways almost entirely.”
The pathways they detected were glycolysis and the pentose phosphate pathway, “reactions that form the core metabolic backbone of every living cell,” Ralser adds. Together these pathways produce some of the most important materials in modern cells, including ATP – the molecule cells use to drive their machinery, the sugars that form DNA and RNA, and the molecules needed to make fats and proteins.
If these metabolic pathways were occurring in the early oceans, then the first cells could have enveloped them as they developed membranes.
In all, 29 metabolism-like chemical reactions were spotted, seemingly catalysed by iron and other metals that would have been found in early ocean sediments. The metabolic pathways aren’t identical to modern ones; some of the chemicals made by intermediate steps weren’t detected. However, “if you compare them side by side it is the same structure and many of the same molecules are formed,” Ralser says. These pathways could have been refined and improved once enzymes evolved within cells.
Reversible reaction
Detecting the metabolite ribose 5-phosphate is particularly noteworthy, Ralser says. This is because it is a precursor to RNA, which encodes information, catalyses chemical reactions and most importantly of all, can replicate.
“I think this paper has really interesting connotations for the origins of life,” says Matthew Powner at University College London. It hints at how more complex enzymes could have evolved, he says, because substances that made these early processes more efficient would have been selected for.
There is one big problem, however. “For origins of life, it is important to understand where the source molecules come from,” Powner says. No one has yet shown that such substances could form spontaneously in the early oceans.
A related issue is that the reactions observed so far only go in one direction; from complex sugars to simpler molecules like pyruvate. “Given the data, one might well conclude that any organics in the ocean would have been totally degraded, rather than forming the basis of modern metabolism,” says Jack Szostak, who studies the origin of life at Harvard. “I would conclude that metabolism had to evolve, within cells, one reaction and one catalyst at a time.”
But Ralser disagrees. In his opinion, whether the reaction is catalysed by an enzyme or by a molecule in the Archean Ocean leads to the same result; “every chemical reaction is in principle reversible, whether an enzyme or a simple molecule is the catalyst,” he says.
Journal reference: Molecular Systems Biology, DOI: 10.1002/msb.20145228
Read more: Spark of life: Metabolism appears in lab without cells
How did the very first atom come about?
How did the very first gods come about?
Me neither. Good morning indeependent! Have you had your proteins yet?
Indeependent
Diamond Member
- Nov 19, 2013
- 73,633
- 28,506
- 2,250
BINGO!As in rice and beans? So much better than the cyanide (HCN) that Miller produced.My wife and I became vegetarians and we love our proteins!Me nei
I don't believe in man made gods that need me to burn my child on a pyre.The magic of the gods.Of course these studies omit how the atoms from which these atoms came about.Yet more evidence that creationists / supernaturalists are making appeals to gods who are not required for existence.

Spark of life: Metabolism appears in lab without cells
Complex processes that support all life have been serendipitously spotted with no cells needed, hinting that life could have started spontaneously in early oceanswww.newscientist.com
Metabolic processes that underpin life on Earth have arisen spontaneously outside of cells. The serendipitous finding that metabolism – the cascade of reactions in all cells that provides them with the raw materials they need to survive – can happen in such simple conditions provides fresh insights into how the first life formed. It also suggests that the complex processes needed for life may have surprisingly humble origins.
“People have said that these pathways look so complex they couldn’t form by environmental chemistry alone,” says Markus Ralser at the University of Cambridge who supervised the research.
But his findings suggest that many of these reactions could have occurred spontaneously in Earth’s early oceans, catalysed by metal ions rather than the enzymes that drive them in cells today.
The origin of metabolism is a major gap in our understanding of the emergence of life. “If you look at many different organisms from around the world, this network of reactions always looks very similar, suggesting that it must have come into place very early on in evolution, but no one knew precisely when or how,” says Ralser.
Happy accident
One theory is that RNA was the first building block of life because it helps to produce the enzymes that could catalyse complex sequences of reactions. Another possibility is that metabolism came first; perhaps even generating the molecules needed to make RNA, and that cells later incorporated these processes – but there was little evidence to support this.
“This is the first experiment showing that it is possible to create metabolic networks in the absence of RNA,” Ralser says.
Remarkably, the discovery was an accident, stumbled on during routine quality control testing of the medium used to culture cells at Ralser’s laboratory. As a shortcut, one of his students decided to run unused media through a mass spectrometer, which spotted a signal for pyruvate – an end product of a metabolic pathway called glycolysis.
To test whether the same processes could have helped spark life on Earth, they approached colleagues in the Earth sciences department who had been working on reconstructing the chemistry of the Archean Ocean, which covered the planet almost 4 billion years ago. This was an oxygen-free world, predating photosynthesis, when the waters were rich in iron, as well as other metals and phosphate. All these substances could potentially facilitate chemical reactions like the ones seen in modern cells.
Metabolic backbone
Ralser’s team took early ocean solutions and added substances known to be starting points for modern metabolic pathways, before heating the samples to between 50 ˚C and 70 ˚C – the sort of temperatures you might have found near a hydrothermal vent – for 5 hours. Ralser then analysed the solutions to see what molecules were present.
“In the beginning we had hoped to find one reaction or two maybe, but the results were amazing,” says Ralser. “We could reconstruct two metabolic pathways almost entirely.”
The pathways they detected were glycolysis and the pentose phosphate pathway, “reactions that form the core metabolic backbone of every living cell,” Ralser adds. Together these pathways produce some of the most important materials in modern cells, including ATP – the molecule cells use to drive their machinery, the sugars that form DNA and RNA, and the molecules needed to make fats and proteins.
If these metabolic pathways were occurring in the early oceans, then the first cells could have enveloped them as they developed membranes.
In all, 29 metabolism-like chemical reactions were spotted, seemingly catalysed by iron and other metals that would have been found in early ocean sediments. The metabolic pathways aren’t identical to modern ones; some of the chemicals made by intermediate steps weren’t detected. However, “if you compare them side by side it is the same structure and many of the same molecules are formed,” Ralser says. These pathways could have been refined and improved once enzymes evolved within cells.
Reversible reaction
Detecting the metabolite ribose 5-phosphate is particularly noteworthy, Ralser says. This is because it is a precursor to RNA, which encodes information, catalyses chemical reactions and most importantly of all, can replicate.
“I think this paper has really interesting connotations for the origins of life,” says Matthew Powner at University College London. It hints at how more complex enzymes could have evolved, he says, because substances that made these early processes more efficient would have been selected for.
There is one big problem, however. “For origins of life, it is important to understand where the source molecules come from,” Powner says. No one has yet shown that such substances could form spontaneously in the early oceans.
A related issue is that the reactions observed so far only go in one direction; from complex sugars to simpler molecules like pyruvate. “Given the data, one might well conclude that any organics in the ocean would have been totally degraded, rather than forming the basis of modern metabolism,” says Jack Szostak, who studies the origin of life at Harvard. “I would conclude that metabolism had to evolve, within cells, one reaction and one catalyst at a time.”
But Ralser disagrees. In his opinion, whether the reaction is catalysed by an enzyme or by a molecule in the Archean Ocean leads to the same result; “every chemical reaction is in principle reversible, whether an enzyme or a simple molecule is the catalyst,” he says.
Journal reference: Molecular Systems Biology, DOI: 10.1002/msb.20145228
Read more: Spark of life: Metabolism appears in lab without cells
How did the very first atom come about?
How did the very first gods come about?
Me neither. Good morning indeependent! Have you had your proteins yet?
No Head, No Oil.
Vegetable broth replaced oil.
We're never hungry as we eat all day.
I will admit that if we take a long walk, I can't eat until after the walk because everything goes right through me.
Again, the top 10 amino acids by proportion - only Alanine, Glycine and aspartic acid are in proteins:
Glycine - C₂H₅NO₂ - proportion: 440
Alanine - C3H7NO2 - proportion: 790
alpha-aminobutyric acid - C₄H₉NO₂ - proportion: 270
a(alpha)-Hydroxy-aminobutyric acid - C4H8O3 - proportion: 74
Norvaline - C5H11NO2 - proportion: 61
Sarcosine - C3H7NO2 - proportion: 55
Aspartic acid - C4H7NO4 - proportion: 34
2,4 [alpha/gamma]-diaminobutyric acid- C4H10N2O2 - proportion: 33
alpha[2]-Aminoisobutyric acid - H2N-C(CH3)2-COOH; C₄H₉NO₂ - proportion 30
N-Ethylglycine -C4H9NO2 - proportion: 30
I forgot about alpha-aminobutyric acid (33). It is also not used in proteins. The 20 amino acids used in proteins are:
1. alanine - ala - A
2. arginine - arg - R
3. asparagine - asn - N
4. aspartic acid - asp - D
5. cysteine - cys - C
6. glutamine - gln - Q
7. glutamic acid - glu - E
8. glycine - gly - G
9. histidine - his - H
10. isoleucine - ile - I
11. leucine - leu - L
12. lysine - lys - K
13. methionine - met - M
14. phenylalanine - phe - F
15. proline - pro - P
16. serine - ser - S
17. threonine - thr - T
18. tryptophan - trp - W
19. tyrosine - tyr - Y
20. valine - val - V
Glycine - C₂H₅NO₂ - proportion: 440
Alanine - C3H7NO2 - proportion: 790
alpha-aminobutyric acid - C₄H₉NO₂ - proportion: 270
a(alpha)-Hydroxy-aminobutyric acid - C4H8O3 - proportion: 74
Norvaline - C5H11NO2 - proportion: 61
Sarcosine - C3H7NO2 - proportion: 55
Aspartic acid - C4H7NO4 - proportion: 34
2,4 [alpha/gamma]-diaminobutyric acid- C4H10N2O2 - proportion: 33
alpha[2]-Aminoisobutyric acid - H2N-C(CH3)2-COOH; C₄H₉NO₂ - proportion 30
N-Ethylglycine -C4H9NO2 - proportion: 30
I forgot about alpha-aminobutyric acid (33). It is also not used in proteins. The 20 amino acids used in proteins are:
1. alanine - ala - A
2. arginine - arg - R
3. asparagine - asn - N
4. aspartic acid - asp - D
5. cysteine - cys - C
6. glutamine - gln - Q
7. glutamic acid - glu - E
8. glycine - gly - G
9. histidine - his - H
10. isoleucine - ile - I
11. leucine - leu - L
12. lysine - lys - K
13. methionine - met - M
14. phenylalanine - phe - F
15. proline - pro - P
16. serine - ser - S
17. threonine - thr - T
18. tryptophan - trp - W
19. tyrosine - tyr - Y
20. valine - val - V
I didn't read this the first few times you cut and pasted it.Again, the top 10 amino acids by proportion - only Alanine, Glycine and aspartic acid are in proteins:
Glycine - C₂H₅NO₂ - proportion: 440
Alanine - C3H7NO2 - proportion: 790
alpha-aminobutyric acid - C₄H₉NO₂ - proportion: 270
a(alpha)-Hydroxy-aminobutyric acid - C4H8O3 - proportion: 74
Norvaline - C5H11NO2 - proportion: 61
Sarcosine - C3H7NO2 - proportion: 55
Aspartic acid - C4H7NO4 - proportion: 34
2,4 [alpha/gamma]-diaminobutyric acid- C4H10N2O2 - proportion: 33
alpha[2]-Aminoisobutyric acid - H2N-C(CH3)2-COOH; C₄H₉NO₂ - proportion 30
N-Ethylglycine -C4H9NO2 - proportion: 30
I forgot about alpha-aminobutyric acid (33). It is also not used in proteins. The 20 amino acids used in proteins are:
1. alanine - ala - A
2. arginine - arg - R
3. asparagine - asn - N
4. aspartic acid - asp - D
5. cysteine - cys - C
6. glutamine - gln - Q
7. glutamic acid - glu - E
8. glycine - gly - G
9. histidine - his - H
10. isoleucine - ile - I
11. leucine - leu - L
12. lysine - lys - K
13. methionine - met - M
14. phenylalanine - phe - F
15. proline - pro - P
16. serine - ser - S
17. threonine - thr - T
18. tryptophan - trp - W
19. tyrosine - tyr - Y
20. valine - val - V
Could you cut and paste it again?
BreezeWood
VIP Member
- Oct 26, 2011
- 17,275
- 1,396
- 85
OK. But how does a bunch of inanimate "stuff" make the leap to a living organism? I know all about the building blocks of life. I have yet to see humans take said building blocks and make the blocks start walking around.Macro-evolution (speciation), has a great deal of observational evidence.But you don't love the ones that conflict with the Bible (e.g., evolution and expansion)?You're missing my point...I love theories that drive improvements in science.
Expansion is referred to in Isaiah 40:22 and other similar verses. Micro-evolution is confirmed by the limited size of Noah's ark (albeit very large) which allows for all kinds of animals to have been saved on the Ark - but not all species (e.g. likely 2 cats which have become multiple 'species' of cats) would have fit on the ark.
Micro-evolution is fact - it has been observed in many ways, including newer observations in the field of epigenetics. Macro-evolution is theory which has no observational evidence but just speculation.
False - but you must be thinking of an example - why not post one and enlighten me!Macro-evolution is theory which has no observational evidence but just speculation..False - but you must be thinking of an example - why not post one and enlighten me!
there is abundant evidence for the process for speciation -
View attachment 330677
transforming from one being into another is an everyday occurrence of metamorphosis - the same process would have similar results parent to sibling transformation in a single instance of a new being as a metaphysical exercise developed over numerous generations till its final enactment in a single step and reproduced by the new sibling for all future generations.a living organism is composed of two ingredients - physiology and spiritual content - and the prevailing conditions conducive to their development all of which occurred on planet Earth together and with a precipitous bolt of lightening at the exact time necessary the first cell was born.OK. But how does a bunch of inanimate "stuff" make the leap to a living organism?
Lightning - Miller used electric discharge which is supposed imitate Lightning. However, Miller only got a mixture of mostly non-biologic chemical reaction products - with formic acid the most prevalent by proportion. How do you propose a living cell resulted from that mixture (aka primordial soup)?
Yes, metamorphosis is amazing - but it is NOT speciation - the DNA is the same.
.transforming from one being into another is an everyday occurrence of metamorphosis - the same process would have similar results parent to sibling transformation in a single instance of a new being as a metaphysical exercise developed over numerous generations till its final enactment in a single step and reproduced by the new sibling for all future generations.
you asked for an example for the process of speciation, metamorphosis is the transformation of one being into another w/ you claim the same dna - the transformation is the example you requested for the process of speciation, mutation is not what alters dna that is done by the parent metaphysically to alter their sibling to compliment in some way their survival and when successful is passed to all succeeding generations - speciation is the same process augmented over time for a full implementation to create a new being for the same purpose for the siblings future prosperity.
.Milleronlygot a mixture of (mostly) non-biologic chemical reaction products ...
so there were "biologic chemical reaction products" produced by the experiment - howabout the glass is half full or even .o1 full - to becoming the 1st physiological cell is an advancement that continues to this day.
sorry, an electrode in a beacon is not lightning in the instance of the purity of primordial Earth. please do not refer to the Almighty's creation as a pond of muck.
It might find it shocking to learn that science has accumulated knowledge since Miller-Urey. Unlike the bibles, science knowledge is not static.As noted fatty acids were also produced in Miller's experiment (most sources ignore this). "
a carboxylic acid consisting of a hydrocarbon chain and a terminal carboxyl group ....
a fatty acid is a carboxylic acid with a long aliphatic chain, which is either saturated or unsaturated."
I posted above about carboxylic acids in general. The carboxyl group is a carbon atom double bonded to an Oxygen atom and single bonded to to a hydroxyl group (-OH). A hydrocarbon is a molecule with Hydrogen and Carbon (e.g. Methane = CH4). An aliphatic chain consists of hydrocarbons that form an open chain as in fatty acids and alkanes to be distinguished from hydrocarbon chains which form rings instead of open chains. Alkanes are saturated hydrocarbons like methane (CH4), ethane (C2H6) and propane (C3H8). Saturated hydrocarbons have single bonds between hydrogen atoms - the highest possible number of hydrogen atoms (e.g. methane/CH4) while unsaturated hydrocarbons have double or triple bonds between adjacent carbon atoms and thus can have hydrogen atoms added to make the compound saturated.
More from the Thaxton et al link concerning carboxylic acids from page 55:
"In a similar fashion, growing polypeptides would be terminated by reactions with amines, aldehydes, ketones, reducing sugars* or carboxylic acids. If by some remote chance a true protein did develop in the ocean, its viability would be predictably of short duration. For example, formaldehyde would readily react with free amino groups to form methylene cross-linkages between proteins.37 This would tie up certain reactive sites, and retard the reaction of protein with other chemical agents. To illustrate, "irreversible combination of formaldehyde with asparagine amide groups" would result in a compound which is "stable to dilute boiling phosphoric acid."38 This tying up process is the principle of the well-known tanning reaction, and is used similarly to retard cadaver decay. "In general, reaction with formaldehyde hardens proteins, decreases their water-sensitivity, and increases their resistance to the action of chemical reagents and enzymes."39 Survival of proteins in the soup would have been difficult indeed."
Footnote:
"It is interesting to note that in certain abnormal situations, such as diabetes, the carbonyl group of glucose will form chemical bonds with the amino group of cellular proteins, a process called glycosylation. (See A.L. Notkins, 1975. Sci. Amer. 241,62.)" [Not so good for life!]
Bottom line: carboxylic acids will react with peptides and stop further polymerization. Also the carbonyl group (CO double bonded) in sugars will react with the amino group (NH2) in peptides (and in amines in general). Both sugars (as in RNA) and amines (as in amino acids) are required for life and yet they interact and, in effect: destroy each other!
to be continued later. I'll give you all a chance to review the actual evidence in chemistry relevant to the origin of life and specifically to Miller's experiment.
Again, one needs an intelligent chemist (e.g. God) to create life - and human chemists are inferior creators and cannot create life. Some attribute the attributes of God to Chance! Not a chance!
Actually, we find that supernaturalism is not a requirement for life on the planet.
Before life began on Earth, the environment likely contained a massive number of chemicals that reacted with each other more or less randomly, and it is unclear how the complexity of cells could have emerged from such chemical chaos. Now, a team led by Tony Z. Jia at the Tokyo Institute of Technology and Kuhan Chandru of the National University of Malaysia has shown that simple α-hydroxy acids, like glycolic and lactic acid, spontaneously polymerize and self-assemble into polyester microdroplets when dried at moderate temperatures followed by rehydration. This could be what happened along primitive beaches and river banks, or in drying puddles. These form a new type of cell-like compartment that can trap and concentrate biomolecules like nucleic acids and proteins. These droplets, unlike most modern cells, are able to merge and reform easily, and thus could have hosted versatile early genetic and metabolic systems potentially critical for the origins of life.
All life on Earth is made up of cells. Cells are composed of lipids, proteins and nucleic acids, with the lipid forming the cell membrane, an enclosure that keeps the other components together and interfaces with the environment, exchanging food and waste. How molecular assemblages as complex as cells originally formed remains a mystery.
Most origins of life research focuses on how the molecules and structures of life were produced by the environment, and then assembled into structures that led to the first cells. However, there were likely many other types of molecules that formed alongside biomolecules on early Earth, and it is possible that life started using very simple chemistry unrelated to modern biomolecules, then evolved through increasingly complex stages to give rise to the structures found in modern cells.
Previous work conducted at ELSI showed that moderate temperature drying of the simple organic compounds known as alpha-hydroxy acids, which are found in meteorites and many simulations of prebiological chemistry, spontaneously polymerizes them into mixtures of long polyesters. Building on this work, Jia and colleagues took the next step and examined these reactions under the microscope, and found that these mixed polyester systems form a gel phase and spontaneously self-assemble when rewetted to form simple cell-like structures.
The most challenging aspect of this work was devising new methods to characterize the droplets' properties and functions, as no one had analyzed such systems before. Jia noted that the team was fortunate to have such a diversity of multidisciplinary expertise, including chemists, biochemists, materials scientists and geologists. After determining their composition and showing their propensity to self-assemble, the next question was whether these cell-like structures might be able to do something chemically useful. Modern cell membranes perform many crucial functions that help maintain the cell, for example, retaining macromolecules and metabolites in one place, as well as providing a constant internal environment, which can be very different from the one outside the cell. They first measured how stable these structures were and found they could persist for very long periods depending on the environmental conditions, but could also be made to merge and coalesce.
They then tested the ability of these structures to sequester molecules from the environment and found they accumulated large dye molecules to a remarkable degree. They then showed that these droplets could also host RNA and protein molecules and still permit them to be functionally catalytic. Further, the team showed that the droplets could assist in the formation of a lipid layer on their surface, suggesting they could have helped scaffold protocell formation.
Jia and colleagues are not certain these structures are the direct ancestors of cells, but they think it is possible such droplets could have enabled the assembly of protocells on Earth. The new compartmentalization system they have found is extremely simple, they note, and could form easily in primitive environments throughout the universe. Says Jia, "This allows us to imagine non-biological systems on early Earth that could still have had a hand in the origins of life. This suggests there may be many other non-biological systems that should be targets of future investigations of this type." He thinks the development of these or similar model systems could allow better study of the evolution of diverse chemical systems representative of the complex chemistries likely to be found on primitive planetary bodies.
"The early Earth was certainly a messy place chemically," Jia explains, "and often, most origins of life studies focus on modern biomolecules under relatively 'clean' conditions. Perhaps it is important to take these 'messy' mixtures and see if there are interesting functions or structures that can arise from them spontaneously." The authors now think that by systematically increasing the chemical complexity of such systems, they will be able to observe how they evolve over time and possibly discover divergent and emergent properties.
"We have this new experimental system we can now play with, so we can start to study phenomena like evolution and evolvability of these droplets. The possible combinations of structures or functions these droplets might have are almost endless. If the physical rules that govern the formation of droplets are fairly universal in nature, then we hope to study similar systems to discover whether they also can form microdroplets with novel properties," adds Jia.
Finally, while the team is presently focused on understanding the origins of life, they note this basic research could have applications in other areas, for example, drug delivery and personalized medicine. "This is just a wonderful example of the unexpected ways projects can develop when a team of diverse scientists from around the world come together to try and understand new and interesting phenomena," said team member Jim Cleaves, also of ELSI.

Scientists discover new chemistry that may help explain the origins of cellular life
Before life began on Earth, the environment likely contained a massive number of chemicals that reacted with each other more or less randomly, and it is unclear how the complexity of cells could have emerged from such chemical chaos. Now, a team led by Tony Z. Jia at the Tokyo Institute of...phys.org
Hollie - thank you for the link - it does not address the amino acids and proportions in Miller's experiment - in fact, it is research on how a cell membrane could have been synthesized, not how proteins could be created (human chemists LI cannot do this, btw).
Long cut and paste - did you understand any of it?
Your link notes:
"Cells are composed of lipids, proteins and nucleic acids, with the lipid forming the cell membrane, an enclosure that keeps the other components together and interfaces with the environment, exchanging food and waste. How molecular assemblages as complex as cells originally formed remains a mystery."
Note also that your link does not state details about the environment needed to form simple alpha-hydroxy acids nor the chemcal reactions required for their formation. And the membrane produced does not perform the complex functions actual living cells require to be living cells rather than dead cells.
Are you familiar with how complex the simplest living cell membranes are? Like how food and waste exchanging could be created in a cell membrane?
The skill and wisdom involved to create such a cell membrane requires a superior chemist. Not to mention forming the informational molecules necessary for life and getting them, and only them, inside this cell membrane enclosure!
Indeependent
Diamond Member
- Nov 19, 2013
- 73,633
- 28,506
- 2,250
"science knowledge is not static"It might find it shocking to learn that science has accumulated knowledge since Miller-Urey. Unlike the bibles, science knowledge is not static.As noted fatty acids were also produced in Miller's experiment (most sources ignore this). "
a carboxylic acid consisting of a hydrocarbon chain and a terminal carboxyl group ....
a fatty acid is a carboxylic acid with a long aliphatic chain, which is either saturated or unsaturated."
I posted above about carboxylic acids in general. The carboxyl group is a carbon atom double bonded to an Oxygen atom and single bonded to to a hydroxyl group (-OH). A hydrocarbon is a molecule with Hydrogen and Carbon (e.g. Methane = CH4). An aliphatic chain consists of hydrocarbons that form an open chain as in fatty acids and alkanes to be distinguished from hydrocarbon chains which form rings instead of open chains. Alkanes are saturated hydrocarbons like methane (CH4), ethane (C2H6) and propane (C3H8). Saturated hydrocarbons have single bonds between hydrogen atoms - the highest possible number of hydrogen atoms (e.g. methane/CH4) while unsaturated hydrocarbons have double or triple bonds between adjacent carbon atoms and thus can have hydrogen atoms added to make the compound saturated.
More from the Thaxton et al link concerning carboxylic acids from page 55:
"In a similar fashion, growing polypeptides would be terminated by reactions with amines, aldehydes, ketones, reducing sugars* or carboxylic acids. If by some remote chance a true protein did develop in the ocean, its viability would be predictably of short duration. For example, formaldehyde would readily react with free amino groups to form methylene cross-linkages between proteins.37 This would tie up certain reactive sites, and retard the reaction of protein with other chemical agents. To illustrate, "irreversible combination of formaldehyde with asparagine amide groups" would result in a compound which is "stable to dilute boiling phosphoric acid."38 This tying up process is the principle of the well-known tanning reaction, and is used similarly to retard cadaver decay. "In general, reaction with formaldehyde hardens proteins, decreases their water-sensitivity, and increases their resistance to the action of chemical reagents and enzymes."39 Survival of proteins in the soup would have been difficult indeed."
Footnote:
"It is interesting to note that in certain abnormal situations, such as diabetes, the carbonyl group of glucose will form chemical bonds with the amino group of cellular proteins, a process called glycosylation. (See A.L. Notkins, 1975. Sci. Amer. 241,62.)" [Not so good for life!]
Bottom line: carboxylic acids will react with peptides and stop further polymerization. Also the carbonyl group (CO double bonded) in sugars will react with the amino group (NH2) in peptides (and in amines in general). Both sugars (as in RNA) and amines (as in amino acids) are required for life and yet they interact and, in effect: destroy each other!
to be continued later. I'll give you all a chance to review the actual evidence in chemistry relevant to the origin of life and specifically to Miller's experiment.
Again, one needs an intelligent chemist (e.g. God) to create life - and human chemists are inferior creators and cannot create life. Some attribute the attributes of God to Chance! Not a chance!
Actually, we find that supernaturalism is not a requirement for life on the planet.
Before life began on Earth, the environment likely contained a massive number of chemicals that reacted with each other more or less randomly, and it is unclear how the complexity of cells could have emerged from such chemical chaos. Now, a team led by Tony Z. Jia at the Tokyo Institute of Technology and Kuhan Chandru of the National University of Malaysia has shown that simple α-hydroxy acids, like glycolic and lactic acid, spontaneously polymerize and self-assemble into polyester microdroplets when dried at moderate temperatures followed by rehydration. This could be what happened along primitive beaches and river banks, or in drying puddles. These form a new type of cell-like compartment that can trap and concentrate biomolecules like nucleic acids and proteins. These droplets, unlike most modern cells, are able to merge and reform easily, and thus could have hosted versatile early genetic and metabolic systems potentially critical for the origins of life.
All life on Earth is made up of cells. Cells are composed of lipids, proteins and nucleic acids, with the lipid forming the cell membrane, an enclosure that keeps the other components together and interfaces with the environment, exchanging food and waste. How molecular assemblages as complex as cells originally formed remains a mystery.
Most origins of life research focuses on how the molecules and structures of life were produced by the environment, and then assembled into structures that led to the first cells. However, there were likely many other types of molecules that formed alongside biomolecules on early Earth, and it is possible that life started using very simple chemistry unrelated to modern biomolecules, then evolved through increasingly complex stages to give rise to the structures found in modern cells.
Previous work conducted at ELSI showed that moderate temperature drying of the simple organic compounds known as alpha-hydroxy acids, which are found in meteorites and many simulations of prebiological chemistry, spontaneously polymerizes them into mixtures of long polyesters. Building on this work, Jia and colleagues took the next step and examined these reactions under the microscope, and found that these mixed polyester systems form a gel phase and spontaneously self-assemble when rewetted to form simple cell-like structures.
The most challenging aspect of this work was devising new methods to characterize the droplets' properties and functions, as no one had analyzed such systems before. Jia noted that the team was fortunate to have such a diversity of multidisciplinary expertise, including chemists, biochemists, materials scientists and geologists. After determining their composition and showing their propensity to self-assemble, the next question was whether these cell-like structures might be able to do something chemically useful. Modern cell membranes perform many crucial functions that help maintain the cell, for example, retaining macromolecules and metabolites in one place, as well as providing a constant internal environment, which can be very different from the one outside the cell. They first measured how stable these structures were and found they could persist for very long periods depending on the environmental conditions, but could also be made to merge and coalesce.
They then tested the ability of these structures to sequester molecules from the environment and found they accumulated large dye molecules to a remarkable degree. They then showed that these droplets could also host RNA and protein molecules and still permit them to be functionally catalytic. Further, the team showed that the droplets could assist in the formation of a lipid layer on their surface, suggesting they could have helped scaffold protocell formation.
Jia and colleagues are not certain these structures are the direct ancestors of cells, but they think it is possible such droplets could have enabled the assembly of protocells on Earth. The new compartmentalization system they have found is extremely simple, they note, and could form easily in primitive environments throughout the universe. Says Jia, "This allows us to imagine non-biological systems on early Earth that could still have had a hand in the origins of life. This suggests there may be many other non-biological systems that should be targets of future investigations of this type." He thinks the development of these or similar model systems could allow better study of the evolution of diverse chemical systems representative of the complex chemistries likely to be found on primitive planetary bodies.
"The early Earth was certainly a messy place chemically," Jia explains, "and often, most origins of life studies focus on modern biomolecules under relatively 'clean' conditions. Perhaps it is important to take these 'messy' mixtures and see if there are interesting functions or structures that can arise from them spontaneously." The authors now think that by systematically increasing the chemical complexity of such systems, they will be able to observe how they evolve over time and possibly discover divergent and emergent properties.
"We have this new experimental system we can now play with, so we can start to study phenomena like evolution and evolvability of these droplets. The possible combinations of structures or functions these droplets might have are almost endless. If the physical rules that govern the formation of droplets are fairly universal in nature, then we hope to study similar systems to discover whether they also can form microdroplets with novel properties," adds Jia.
Finally, while the team is presently focused on understanding the origins of life, they note this basic research could have applications in other areas, for example, drug delivery and personalized medicine. "This is just a wonderful example of the unexpected ways projects can develop when a team of diverse scientists from around the world come together to try and understand new and interesting phenomena," said team member Jim Cleaves, also of ELSI.

Scientists discover new chemistry that may help explain the origins of cellular life
Before life began on Earth, the environment likely contained a massive number of chemicals that reacted with each other more or less randomly, and it is unclear how the complexity of cells could have emerged from such chemical chaos. Now, a team led by Tony Z. Jia at the Tokyo Institute of...phys.org
Hollie - thank you for the link - it does not address the amino acids and proportions in Miller's experiment - in fact, it is research on how a cell membrane could have been synthesized, not how proteins could be created (human chemists LI cannot do this, btw).
Long cut and paste - did you understand any of it?
Your link notes:
"Cells are composed of lipids, proteins and nucleic acids, with the lipid forming the cell membrane, an enclosure that keeps the other components together and interfaces with the environment, exchanging food and waste. How molecular assemblages as complex as cells originally formed remains a mystery."
Note also that your link does not state details about the environment needed to form simple alpha-hydroxy acids nor the chemcal reactions required for their formation. And the membrane produced does not perform the complex functions actual living cells require to be living cells rather than dead cells.
Are you familiar with how complex the simplest living cell membranes are? Like how food and waste exchanging could be created in a cell membrane?
The skill and wisdom involved to create such a cell membrane requires a superior chemist. Not to mention forming the informational molecules necessary for life and getting them, and only them, inside this cell membrane enclosure!
Correct!
Now where did the first particle come from?
Similar threads
- Replies
- 174
- Views
- 762
- Replies
- 210
- Views
- 1K
- Replies
- 311
- Views
- 3K
Latest Discussions
- Replies
- 223
- Views
- 985
- Replies
- 98
- Views
- 702
- Replies
- 121
- Views
- 556
- Replies
- 20
- Views
- 51
Forum List
-
-
-
-
-
Political Satire 8526
-
-
-
-
-
-
-
-
-
-
-
-
-
-
-
-
-
-
-
ObamaCare 781
-
-
-
-
-
-
-
-
-
-
-
Member Usernotes 485
-
-
-
-
-
-
-
-
-
-